Detecting Dementia
Looking to the retina to find a biomarker for frontotemporal degeneration
Everyone is familiar with Alzheimer’s disease (AD) – it’s the most common cause of dementia (1). But few are familiar with frontotemporal degeneration (FTD); another dementia that’s characterized by neuronal atrophy in the frontal and temporal lobes. Despite its relative obscurity, it’s almost as common as AD in people under the age of 65, but it is poorly understood (2).

Certain forms of FTD share a characteristic with AD – tauopathies, or the accumulation of tau protein. Tau accumulates and forms inclusion bodies in neurons, resulting in neurofibrillary tangles that lead to cellular dysfunction and eventual cell death.
“There is an urgent need to find biomarkers for patients with FTD who have a tauopathy,” says Benjamin Kim, assistant professor of Ophthalmology at University of Pennsylvania’s Scheie Eye Institute, USA. Inspired to find a solution, Kim and his team turned to data published by a fellow lab at the University of Pennsylvania that showed that mice with a mutation in the microtubule-associated protein Rp1 gene had photoreceptor abnormalities that were detectable by OCT (3). “Proper microtubule function is critical for photoreceptors,” says Kim. “Because tau is also a microtubule-associated protein – which evidence suggests is also expressed in photoreceptors – we hypothesized that patients with a tauopathy might also have photoreceptor abnormalities detectable by OCT.”
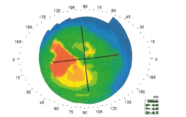
In their cross-sectional study, they examined the retinae of 27 patients with FTD (46 eyes) and 44 controls (69 eyes) using spectral-domain OCT (SD-OCT) (4). Of those with FTD, 19 (31 eyes) were identified as having probable tauopathy based on cerebrospinal fluid (CSF), genetic and clinical analyses. What structural differences did they find? Significantly thinner outer retinal layers (ORL) in patients with FTD compared with controls (132 vs. 142 µm, p=0.004), as well as significantly thinner outer nuclear layers (ONL; 88.5 vs. 97.9 µm, p=0.0030 and ellipsoid zones (EZ; 14.5 vs. 15.1 µm, p=0.009); patients in the tauopathy subgroup also exhibited thinning of these layers (p=0.01). The group also identified an association between the degree of thinning and results from MMSE (a screening examination for dementia), suggesting that outer retina thinning may correlate with disease severity.
“Our results of photoreceptor thinning were in line with our hypothesis, but it was surprising that there was no inner retinal thinning, as this has been seen in several other neurodegenerative diseases,” says Kim. “We find our data exciting because we are seeing that dementia patients with different brain pathology may also have specific, different retinal abnormalities as well.”
Ultimately, the team hopes that OCT analysis of the retina could be used as a biomarker for patients with FTD who have a tauopathy, but must first learn how OCT findings change over time in relation to disease. “We are now in an era where therapies specific to tau are being tested; if the use of OCT could become a validated biomarker, it would help increase the chance of finding treatments for these patients.”
- Alzheimer’s Association. “2017 Alzheimer’s disease facts and figures”. Available at: bit.ly/ADFacts. Accessed: 15 September, 2017.
- DJ Irwin et al., “Frontotemporal lobar degeneration: defining phenotypic diversity through personalized medicine”, Acta Neuropathol, 129, 469–491 (2015). PMID: 25549971.
- D Song et al., “A murine RP1 missense mutation causes protein mislocalization and slowly progressive photoreceptor degeneration”, Am J Pathol, 184, 2721–2729 (2014). PMID: 25088982.
- BJ Kim et al., “Optical coherence tomography identifies outer retina thinning in frontotemporal degeneration”, Neurology, [Epub ahead of print], (2017). PMID: 28887373.