Demodex blepharitis is a common, but underrecognized condition associated with significant discomfort and reduced quality of life for patients. Many eye care professionals may fail to recognize the prevalence – or the signs and symptoms – of Demodex blepharitis; others may overlook it due to a lack of available treatment options. There is currently no FDA-approved pharmaceutical specifically indicated for this common ocular surface disease (OSD).
We can draw a parallel between our current understanding of the importance of diagnosing and treating Demodex blepharitis and where we were with our recognition of dry eye disease (DED) 20 years ago, or meibomian gland disease (MGD) even more recently. Two decades ago, most ophthalmologists were not diagnosing DED; in fact, many did not believe they saw these patients in their practice at all. It wasn’t until we had a prescription treatment (cyclosporine) and improved diagnostics that we started actively looking for DED – and then we found it everywhere. We realized that this was an inflammatory disease process and that patients benefited from treatment.
As time went on, diagnostic methods further improved and we turned our attention to the meibomian glands. Once we had the tools to identify MGD, we began to understand its prevalence and the importance of managing it to improve the health of the tear film. The development of therapies like thermal pulsation followed. After DED and MGD, we could argue that blepharitis is the next horizon in OSD. Today, we are gaining a deeper understanding of the prevalence and impact of Demodex blepharitis – and how to look for it now that a promising prescription eye drop is waiting in the wings.
Collarettes: pathognomonic sign of Demodex blepharitis. Collarettes have also been referred to as dandruff-like material, cylindrical dandruff, scurf, and crust. Though Demodex mites are common on skin, the presence of collarettes indicates overgrowth. This clear to whitish waxy buildup left by proliferating mites is a combination of partially digested epithelial cells, keratin, waste, and mite eggs. The cuffs are typically seen at the base of the eyelashes, particularly on the upper lid. Collarettes are best observed on the upper eyelid when patients are looking down during a slit-lamp exam.
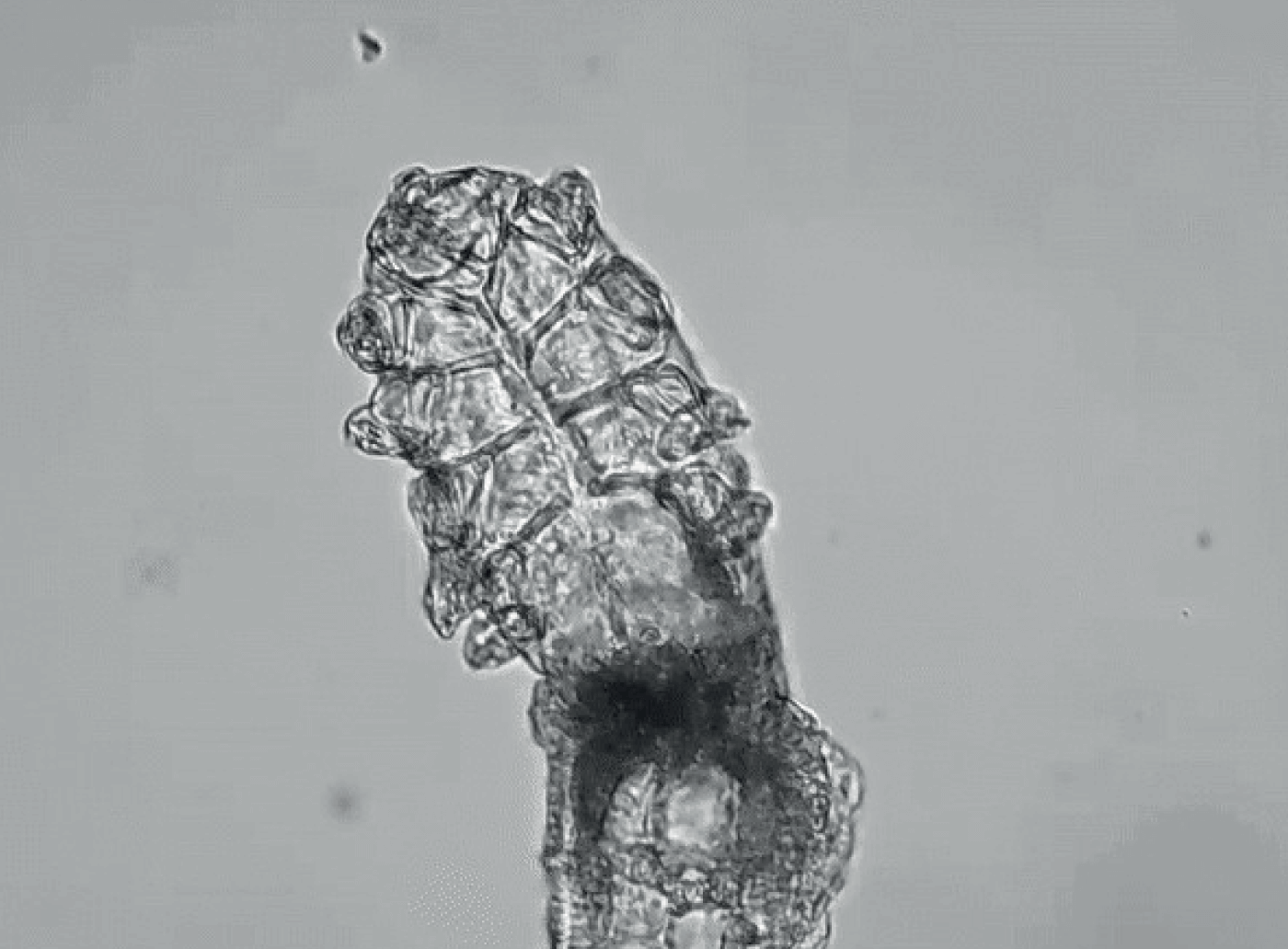
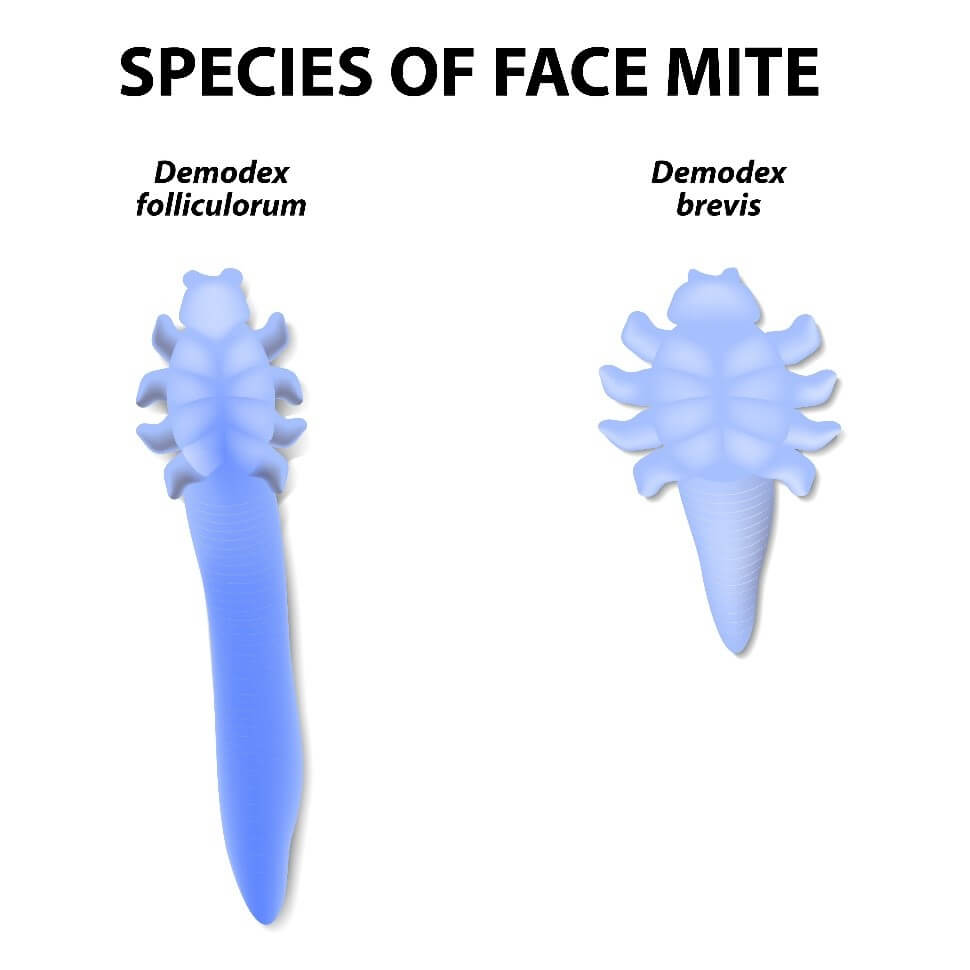
Demodex folliculorum: larger species of Demodex mite that burrows into eyelash follicles.
Demodex brevis: smaller species of Demodex mite that inhabits the sebaceous and meibomian glands.
Condition prevalence
Blepharitis is a chronic, progressive condition characterized by inflammation, ocular irritation, and erythema (1). Extrapolations based on population and prevalence suggest that up to 20 million US adults may have blepharitis. When left untreated, the condition can lead to significant sequelae. Although blepharitis primarily affects the lid margin, it can affect the eyelid skin, base of the eyelashes, eyelash follicles, and the meibomian glands and gland orifices, which is why patients may present with red, irritated, or itchy eyelids and eyelash debris.
Many factors can be implicated in blepharitis; however, studies exploring the association of blepharitis and Demodex infestation have shown a significant correlation between the two. In a meta-analysis of 13 controlled studies, it was reported that Demodex infestation occurs in 44.5 percent of blepharitis subjects versus 16.7 percent in normal controls (2). Biernat et al. also reported that the prevalence of ocular Demodex infestation is significantly correlated with blepharitis (3).
Demodex mites are the most common ectoparasites found on humans. There are two species of Demodex – D. folliculorum and D. brevis – that live on the skin of the face and eyelids. The former burrows into eyelash follicles, whereas the latter inhabits the sebaceous and meibomian glands (4, 5). Although Demodex mites are common in low numbers, an overpopulation or infestation of mites, also called demodicosis, is associated with blepharitis (6). Demodex infestation increases with age (3,7, 8, 9), present in 84 percent of patients 60 and older and in nearly all patients by the time they reach 70 (8).
Recent observations reveal just how common Demodex infestation is. A multicenter observational trial in eight ophthalmology and optometry practices quantified how many patients – out of sequential patients presenting for any purpose – had collarettes, a pathognomonic sign of Demodex infestation (10). Of 1,121 patients examined, 58 percent had collarettes – a proportion that translates to 25 million people in the United States who may have Demodex blepharitis (10).
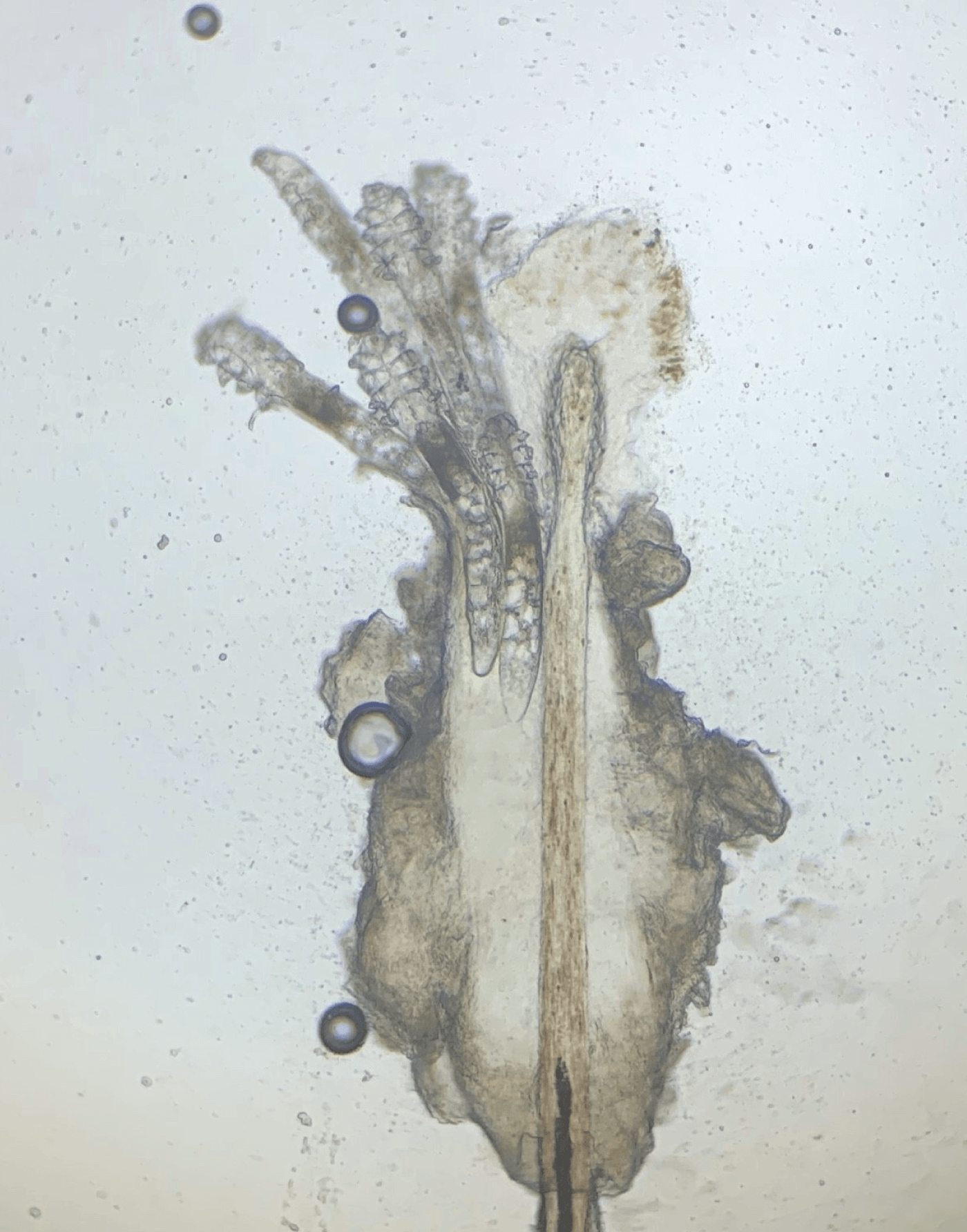
Demodex mites contribute to blepharitis inflammation via mechanical, chemical, and bacterial mechanisms (5). The overcrowded mites scrape the epithelial cell lining with their claws and lay eggs in the follicle, causing follicular distention, misdirected lashes, madarosis, and irritation. Mites also excrete digestive enzymes as they feed and exude waste when they die, which causes inflammation, hyperemia, irritation, and epithelial hyperplasia. Lastly, bacteria living on the mite surface or in its gut cause inflammation of the surrounding ocular tissues. As the mites proliferate in this nutrient-rich environment, the partially digested epithelial cells, waste, and eggs form collarettes (or “cylindrical dandruff”) at the base of the lashes. These collarettes are now recognized as pathognomonic for Demodex blepharitis (9, 11).
There are currently no FDA-approved treatments for blepharitis caused by Demodex infestation. The American Academy of Ophthalmology’s Preferred Practice Pattern (PPP) for blepharitis suggests a combination of antibiotics, topical anti-inflammatory agents, and daily lid hygiene (1). Though these therapies can help manage the symptoms, to address the root cause of the disease, the mite must be eradicated.
The most common lid hygiene approach involves the use of scrubs, wipes, or gels containing tea tree oil (TTO) or its major acaricidal component, terpinen-4-ol (T4O). TTO and T4O products are made in a variety of concentrations and many have been tested. Most studies, however, were small and not placebo-controlled (12, 13, 14, 15, 16), and a recent Cochrane review found mixed evidence of their efficacy (17). Other work reveals that TTO can be toxic to epithelial cells and fibroblasts (18, 19, 20).
Clinical significance
Many symptoms of ocular surface diseases overlap, making Demodex blepharitis hard to distinguish. Patients may have red eyes and feel discomfort, dryness, itching, or burning. Older patients in particular may not recognize or note any discomfort. But, left unmanaged, Demodex blepharitis can lead to tear film instability with fluctuating and blurred vision, lid and lash abnormalities, inflammation of the conjunctiva and surrounding skin, suboptimal surgical outcomes, contact lens intolerance and reduced wear time, noticeable eye and eyelid erythema, corneal damage, and a reduced quality of life. Clinicians must ask specific questions to elicit more information and to screen for signs of the condition by initiating the key diagnostic steps.
To identify Demodex blepharitis, the physician should have the patient look down to examine the base of the lash on the upper lid. The upper lid is often overlooked and only partially visible when the patient is looking straight ahead. In that position, the base of the lashes is somewhat hidden and collarettes present on the upper lid may be missed. Visualizing the lashes and the upper lid is key to the clinical exam.
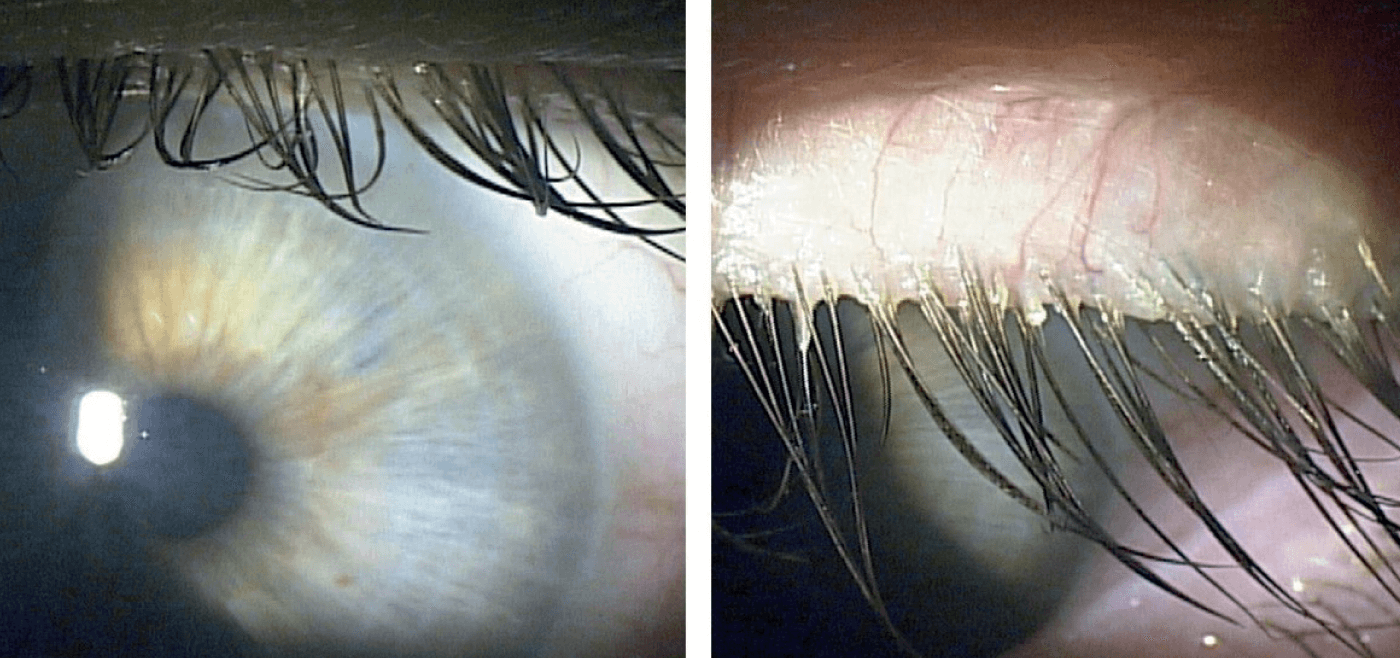
Typically found at the base of the lash, collarettes can migrate up as the hair shaft grows. They appear as clear to whitish waxy cuffs encircling the base of the eyelashes on the upper lid. As mentioned earlier, collarette presence is a sure sign of Demodex (9, 11, 21, 22) – meaning that this straightforward diagnosis may not require epilation and microscopic evaluation of eyelashes. Other signs include eyelash disorders, crusting and redness of the lid margin, and inflammation of the lid margin, conjunctiva, and cornea (blepharoconjunctivitis and blepharokeratitis).
I educate patients about Demodex mites and their high prevalence on human skin. I explain that they have a mite overgrowth that I have identified by observing debris on the base of their lashes. I let them know that we will develop a treatment plan together. I also inform them that, in the near future, we hope to have a drop that eradicates Demodex mites.
Clinical approach
My treatment approach starts with teaching patients about lid hygiene, using lid scrubs, and washing the lashes. I recommend various lid wipes that are available over the counter. For patients with extensive collarettes, I clean the biofilm from the lid margin and lashes using microblepharoexfoliation. In my algorithm, the final step would be TTO. As mentioned earlier, it is not without its drawbacks, but there is some efficacy as well (23, 24, 25).
Demodex blepharitis patients may have an MGD component that should be managed concurrently. In those cases, I use topical azithromycin or sometimes an antibiotic-steroid ointment such as Tobradex (tobramycin plus dexamethasone). I favor thermal pulsation to heat the lids and then express the glands. In cases of chronic MGD, I consider oral doxycycline. Doxycycline or minocycline is also very effective for rosacea, which often coexists in these patients.
Compliance with lid hygiene can be a challenge. The more symptomatic patients are, the more likely they are to adhere to their treatments. For many patients, redness and other signs of Demodex blepharitis are a psychosocial concern, so I let them know that the more faithful they are to the lid hygiene protocol – morning and evening –the better their eyelids will look (and feel).
The new ocular surface frontier
With DED, it took an FDA-approved prescription drug to set in motion a sea change in our appreciation for the disease. With MGD, it took enhanced diagnostic tools and approved treatment devices. Now, we recognize the prevalence and impact of these diseases – and the importance of treatment to improve patient comfort, visual satisfaction, and overall quality of life. I see Demodex blepharitis as the next ocular surface disease to follow that trajectory. We have learned how prevalent it is and how to diagnose it. Soon, clinicians may have the first FDA-approved prescription drop specifically indicated for the treatment of Demodex blepharitis in their toolkit.
Tarsus Pharmaceuticals, Inc., is developing a novel therapeutic –TP-03 – based on the drug lotilaner. The new topical drop in development is designed to paralyze and eradicate Demodex mites through the inhibition of parasite-specific GABA-Cl channels (26). Preclinical ex vivo testing demonstrated that lotilaner ophthalmic solution, 0.25%, killed over 95 percent of Demodex mites within 24 hours (27). The company has completed four Phase II clinical trials, two of which were randomized, controlled studies (28, 29, 30). Key efficacy endpoints for the first two, Phase IIa Mars and Phase IIb Jupiter (28, 29) were collarette grade and mite density, and key efficacy endpoints for the next two, Phase IIa Io and Phase IIb Europa, included collarette cure rate (proportion of patients reaching 0–2 collarettes) and mite eradication rate (proportion of patients reaching 0 mites) (30). TP-03 met its primary and secondary endpoints and was well tolerated throughout.
Clinical trials
Phase II studies Mars and Jupiter have shown that TP-03 is well tolerated and effective at reducing collarettes and Demodex density with 28 days of treatment and maintaining results through at least 90 days (28,29). The two studies evaluated a total of 75 patients, showing statistically significant decreases in collarettes and Demodex density as early as day 14 of treatment. No treatment-related adverse events were observed and patients reported that the drop was comfortable.
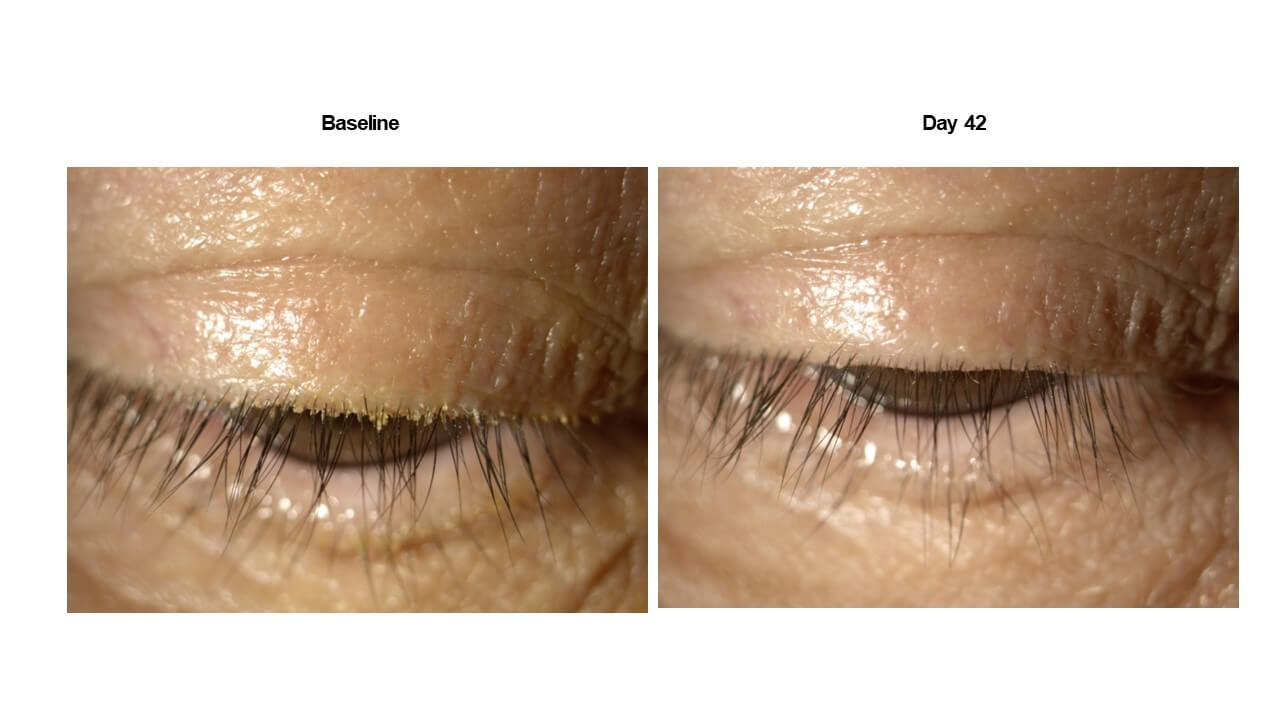
These results were further validated in the Phase IIa Io and Phase IIb Europa studies that included a total of 72 patients (30, 31). In the single-arm open-label trial, Io, TP-03 was effective at achieving collarette cure in 72 percent and mite eradication in 78 percent of patients at day 42. The randomized vehicle-controlled Europa study achieved statistically significant results for collarette cure in 80 percent of participants on TP-03 compared to 16 percent on vehicle (p<.001) at day 42, and mite eradication in 73 percent of participants on TP-03 compared to 21 percent on vehicle (p=.003) at day 42. TP-03 was well tolerated and there were no reports of serious adverse events or treatment discontinuations due to adverse events in either study. Participants in Europa rated the eye drops as “neither comfortable nor uncomfortable,” “comfortable,” or “very comfortable” 87 percent of the time.
Tarsus is now evaluating TP-03 in two pivotal registration trials, Saturn-1 and Saturn-2, the former of which has completed enrollment and the latter of which will begin in Q2 of 2021 (30). The company expects the data will support the potential submission of an NDA for TP-03 for the treatment of Demodex blepharitis, meaning that TP-03 has the potential to be the first FDA-approved therapeutic for this purpose.
Next steps
Demodex blepharitis is a highly prevalent disease that has the potential to cause significant sequelae. The current treatments have some efficacy, but are not validated, often uncomfortable, and do not target the root cause of the disease. Blepharoexfoliation – which needs to be repeated – is the most effective approach for patients with severe collarettes. Some patients who are very vigorous with their lid hygiene may be able to keep their lids clean, but Demodex is a recurrent problem.
Based on the mite eradication and collarette cure rates I have seen reported with TP-03, the drop will move to my initial therapy when available. As I gain clinical experience, I will decide whether or not I need to add other treatments. Excellent efficacy was seen in the clinical trials with lotilaner 0.25% drops. This new treatment has the potential to be first-line therapy for all clinicians.
References
- Cornea/External Disease Panel, “Preferred Practice Pattern Guidelines, Blepharitis” presented at American Academy of Ophthalmology 2018; San Francisco, USA.
- Y-E Zhao et al., “Association of blepharitis with Demodex: A meta-analysis. Ophthalmic Epidemiol”, 19, 95-102 (2012). PMID: 22364595.
- M Biernat et al., “Occurrence of Demodex species in patients with blepharitis and in healthy individuals: A 10-year observational study”, Jpn J Ophthalmol, 62, 628 (2018). PMID: 30255395.
- A Cheng et al., “Recent advances on ocular Demodex infestation”, Curr Opin Ophthalmol., 26, 295 (2015). PMID: 26058028.
- S Fromstein et al., “Demodex blepharitis: Clinical perspectives”, Clin Optom (Auckl), 10, 57 (2018). PMID: 30214343.
- X Luo et al., “Ocular demodicosis as a potential cause of ocular surface inflammation”, Cornea, 36, S9 (2017). PMID: 28902017.
- C Post et al., “E. Demodex folliculorum and blepharitis”, Arch Dermatol., 88, 298 (1963).
- J Liu et al., “Pathogenic role of Demodex mites in blepharitis”, Curr Opin Allergy Clin Immunol, 10, 505 (2010). PMID: 20689407.
- J Zhong et al., “The prevalence of Demodex folliculorum and Demodex brevis in cylindrical dandruff patients”, J Ophthalmol, 8949683 (2019). PMID: 31073414
- Tarsus Collarette Prevalence Study (2020).
- Y-Y Gao et al., “High prevalence of Demodex in eyelashes with cylindrical dandruff”, Invest Ophthalmol Vis Sci, 46, 3089 (2005). PMID: 16123406.
- M Arrúa et al., “Comparative study of the efficacy of different treatment options in patients with chronic blepharitis”, Arch Soc Esp Oftalmol Engl Ed, 90, 112 (2015). PMID: 25542616.
- O Murphy et al., “The efficacy of tea tree face wash, 1,2-octanediol and microblepharoexfoliation in treating Demodex folliculorum blepharitis”, Cont Lens Anterior Eye, 41, 77 (2018). PMID: 29074306.
- Y-Y Gao, “In vitro and in vivo killing of ocular Demodex by tea tree oil”, Br J Ophthalmol, 89, 1468 (2005). PMID: 16234455.
- Y Karakurt and E Zeytun, “Evaluation of the efficacy of tea tree oil on the density of Demodex mites (acari: demodicidae) and ocular symptoms in patients with demodectic blepharitis”, J Parasitol, 104, 473 (2018). PMID: 30016200.
- R Messaoud et al., “Improvement in ocular symptoms and signs in patients with Demodex anterior blepharitis using a novel terpinen-4-ol (2.5%) and hyaluronic acid (0.2%) cleansing wipe”, Clin Ophthalmol, 13, 1043 (2019). PMID: 31417235.
- K Savla et al, “Tea tree oil for Demodex blepharitis”, Cochrane Database Syst Rev, 6, CD013333 (2020). PMID: 32589270.
- A Hayes et al., “In vitro cytotoxicity of Australian tea tree oil using human cell lines”, J Essent Oil Res, 9, 575 (1997).
- R Loughlin et al., “Comparison of the cidal activity of tea tree oil and terpinent-4-ol against clinical bacterial skin isolates and human fibroblast cells”, Lett Appl Microbiol, 46, 428 (2018). PMID: 18298453.
- D Chen et al., “Effects of terpinen-4-ol on meibomian gland epithelial cells in vitro”, Cornea, Online ahead of print (2020). PMID: 32947397.
- M Hom et al., “Demodex: clinical cases and diagnostic protocol”, Optom Vis Sci, 90, 198–(2013).
- A Zhang et al., “Ocular Demodex: a systematic review of the clinical literature”, Ophthal Physiol Op, 40, 389 (2020). PMID: 32691894.
- V Navel et al., “Efficacy of treatments for Demodex blepharitis: A systematic review and meta-analysis”, Ocul Surf, 17, 655 (2019). PMID: 31229586.
- S Tighe et al., “Terpinen-4-ol is the most active ingredient of tea tree oil to kill Demodex mites”, Transl Vis Sci Technolo, 2, 2 (2013). PMID: 24349880.
- H Koo et al., “Ocular surface discomfort and Demodex: Effect of tea tree oil eyelid scrub in Demodex blepharitis”, J Korean Med Sci, 27, 1574 (2012). PMID: 23255861.
- Y Ozoe et al., “The antiparasitic isoxazoline A1443 is a potent blocker of insect ligand-gates chloride channels”, Biochem Biophys Res Commun, 391, 744 (2010). PMID: 19944072.
- J Vehige et al., “A new treatment strategy for Demodex infestation using topic antiparasitic isoxazoline drugs: Results of ex vivo testing”, Poster presented at American Academy of Optometry Annual Meeting; October 23, 2019; Orlando, FL, USA.
- R Gonzalez-Salinas et al., “Pilot study to evaluate the safety and efficacy of TP-03 for the treatment of blepharitis due to Demodex infestation (Mars Study)”, Presented at: Association for Research in Vision and Ophthalmology; 2020. #2984-80042.
- M Hom et al., “Randomized controlled trial to evaluate the safety and efficacy of TP-03 for the treatment of blepharitis due to Demodex infestation (Jupiter Study)” Poster presented at Optometry Meeting; 2020.
- PR Newswire (2021).
- Tarsus, Data presented at the Jefferies Virtual Healthcare Conference; November 17-19, 2020; London, UK.
