
Advertisement feature; Collaboration to provide a one-stop-shop for diabetic macular edema (DME) resource.
Funded by Alimera Sciences. Contains promotional information. Please click here to view the prescribing information and adverse event reporting information.
Alimera Sciences has partnered with The Ophthalmologist to provide an online and living repository for DME-specific information. Alimera Sciences will be leading the content development, supported by Hayward Medical Communications. Content will range from conference reports, case studies, literature reviews, videos, interviews and presentations. The focus will be on raising awareness and education in the field of DME and to share information about the use of ILUVIEN® – an intravitreal fluocinolone acetonide implant – in clinical practice. ILUVIEN is developed and licensed by Alimera Sciences for the treatment of vision impairment associated with chronic DME considered insufficiently responsive to available therapies.1
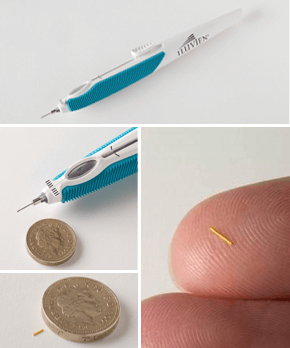
Figure 1. Small enough to be injected into the back of the eye, the ILUVIEN implant is inserted through a 25-gauge needle, using a custom designed, pen-shaped applicator that creates a self-sealing hole upon exit.
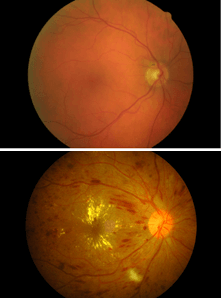
A complication of diabetic retinopathy, DME is a leading cause of blindness,2 with an estimated 21 million people around the globe affected.3 As the prevalence of diabetes continues to grow to over 592 million worldwide within the next generation,4 and with around 6% of diabetics developing the condition,3 DME presents an obvious target for therapeutic intervention. Figure 2. Retinal image of the right eye; showing the macula in a normal healthy eye (top) and DME in a diabetic eye (bottom).
Founded in 2003, Alimera Sciences specializes in the research, development and commercialization of prescription ophthalmic pharmaceuticals.5 ILUVIEN is the first DME treatment to deliver up to 36-months of continuous, low-dose corticosteroid by single injection.5 The drug has since received FDA approval in the United States6 as well as approval in Austria, Denmark, France, Germany, Italy, Norway, Portugal, Spain, Sweden, United Kingdom, Ireland, the Netherlands, Belgium, Luxembourg, Finland, Poland, and the Czech Republic.
Look out for DME content developed by Alimera Sciences on this website throughout 2015. We hope it supports your knowledge of DME and ILUVIEN, and if you would like to contribute material for publication, send your materials to dmecontenthub@hayward.co.uk, we’d be very pleased to consider your contributions.
REFERENCES
1. ILUVIEN SPC. 2013 Available at: www.medicines.org.uk/emc/medicine/27636 (Accessed March 2015)
2. Ford JA, Lois N, Royle P. et al. Current treatments in diabetic macular oedema: systematic review and meta-analysis. BMJ Open. 2013.;3;1-59
3. Yau JW, Rogers SL, Kawasaki R, et al. Global Prevalence and Major Risk Factors Diabetic Retinopathy - For the Meta-Analysis for Eye Disease (META-EYE) Study Group. Diabetes Care. 2012;35:556-564.
4. International Diabetes Atlas. 6th edition: International Diabetes Federation (IDF); 2013. Available at www.idf.org/sites/default/files/EN_6E_Atlas_Full_0.pdf. Accessed March 2015
5. Alimera Sciences. Available at www.alimerasciences.com. Accessed March 2015.
6. New Drug Application for ILUVIEN (fluocinolone acetonide intravitreal insert). Reference NDA 201923. Available at: www.acessdata.fda.gov/drugsatfda_docs/appletter/2014/201923Orig1s000ltr.pdf
UK-ILV-MMM-0169
Date of preparation: March 2015
ILUVIEN® Prescribing Information
Refer to the Summary of Product Characteristics (SPC) before prescribing. Presentation: intravitreal implant in applicator. Each implant contains 190 micrograms of fluocinolone acetonide. Light brown coloured cylinder, approximately 3.5mm x 0.37mm in size. Implant applicator with 25 gauge needle. Indication: ILUVIEN is indicated for the treatment of vision impairment associated with chronic diabetic macular oedema, considered insufficiently responsive to available therapies. Dosage and method of administration: The recommended dose is one ILUVIEN implant in the affected eye. Administration in both eyes concurrently is not recommended. An additional implant may be administered after 12 months if the patient experiences decreased vision or an increase in retinal thickness secondary to recurrent or worsening diabetic macular oedema. Retreatments should not be administered unless the potential benefits outweigh the risks. Only patients who have been insufficiently responsive to prior treatment with laser photocoagulation or other available therapies for diabetic macular oedema should be treated with ILUVIEN. Children under 18: No relevant use. Special populations: No dosage adjustments are necessary in elderly patients, or those with renal or hepatic impairment. Contraindications: the presence of pre-existing glaucoma or active or suspected ocular or periocular infection including most viral diseases of the cornea and conjunctiva, including active epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, varicella, mycobacterial infections, and fungal diseases. Hypersensitivity to the active substance or to any of the excipients. Special warnings and precautions: Intravitreal injections have been associated with endophthalmitis, elevation in intraocular pressure, retinal detachments and vitreous haemorrhages or detachments. Patients should be instructed to report without delay any symptoms suggestive of endophthalmitis. Patient monitoring within two to seven days following the injection may permit early identification and treatment of ocular infection, increase in intraocular pressure or other complication. It is recommended that intra-ocular pressure be monitored at least quarterly thereafter. Use of intravitreal corticosteroids may cause cataracts, increased intraocular pressure, glaucoma and may increase the risk of secondary infections. The safety and efficacy of ILUVIEN administered to both eyes concurrently have not been studied. Concurrent treatment of both eyes is not recommended until the patient’s systemic and ocular response to the first implant is known. Interactions: No interaction studies with other medicinal products have been performed. Pregnancy and lactation: There are no adequate data from the use of intravitreal administered fluocinolone acetonide in pregnant women. ILUVIEN is not recommended during pregnancy unless the potential benefit justifies the potential risk to the foetus. ILUVIEN is not recommended during breast feeding unless clearly necessary. Driving and using machines: Patients may experience temporarily reduced vision after administration of ILUVIEN and should refrain from driving or using machines until this has resolved. Undesirable effects: Very common (≥ 1/10): cataract operation, cataract, increased intraocular pressure, floaters (myodesopsia); common (≥1/100 to < 1/10): glaucoma, trabeculectomy, eye pain, vitreous haemorrhage, conjunctival haemorrhage, blurred vision, glaucoma surgery, reduced visual acuity, vitrectomy, trabeculoplasty; uncommon (≥1/1,000 to < 1/100): endophthalmitis, headache, retinal vascular occlusion, optic nerve disorder, maculopathy, optic atrophy, conjunctival ulcer, iris neovascularisation, retinal exudates, vitreous degeneration, vitreous detachment, posterior capsule opacification, iris adhesions, ocular hyperaemia, sclera thinning, removal of extruded implant from sclera, eye discharge, eye pruritus, extrusion of implant, implant in line of sight, procedural complication, procedural pain. Consult the SPC for full details of undesirable effects. Overdose: No case of overdose has been reported. Legal classification: POM. Pack size and NHS list price: £5,500.00 (ex VAT) for each ILUVIEN 190 micrograms intravitreal implant in applicator. Marketing Authorisation number: PL41472/0001. Marketing Authorisation Holder: Alimera Sciences Limited, Royal Pavilion, Wellesley Road, Aldershot, Hampshire, GU11 1PZ,
United Kingdom. Date of preparation of PI: February 2015
Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard Adverse events should also be reported to Alimera Sciences Limited (telephone: 0800 148 8274) pvalimerasciences@alimerasciences.com
If you are not based in the UK, please refer to your local guidelines for adverse event reporting.
For medical enquiries please email: medicalinformation@alimerasciences.com
Founded in 2003, Alimera Sciences researches and develops innovative vision-improving treatments for chronic retinal disease. Alimera Sciences has developed and licensed ILUVIEN®, an intravitreal implant of 190 micrograms fluocinolone acetonide, for the treatment of vision impairment associated with chronic diabetic macular edema (DME), considered insufficiently responsive to available therapies.a ILUVIEN is the first DME treatment to deliver up to 36 months of continuous, low-dose corticosteroid by single injection.b In 2015, Alimera Sciences has partnered with The Ophthalmologist to facilitate the publication of independently created content on ILUVIEN and DME. Content will range from conference reports, case studies, and literature reviews to video interviews, presentations, and practical information surrounding the use and benefits of ILUVIEN. The word Alimera derives loosely from the Greek, to mean “day of truth”.c With a commitment to honesty, integrity, responsibility, candor, and trust, Alimera Sciences intend that this promotional information accurately and fairly represents the current state of knowledge of ILUVIEN and DME, and is useful to all healthcare professionals involved in DME and its treatment. a. ILUVIEN SPC. 2013 Available at: www.medicines.org.uk/emc/medicine/27636 (Accessed March 2015) b. Alimera Sciences. Available at www.alimerasciences.com (Accessed March 2015) c. Retina Today. 2011. Available at: retinatoday.com/pdfs/0111RT_Wall.Street.pdf (Accessed March 2015) UK-ILV-MMM-0167 Date of preparation: March 2015

