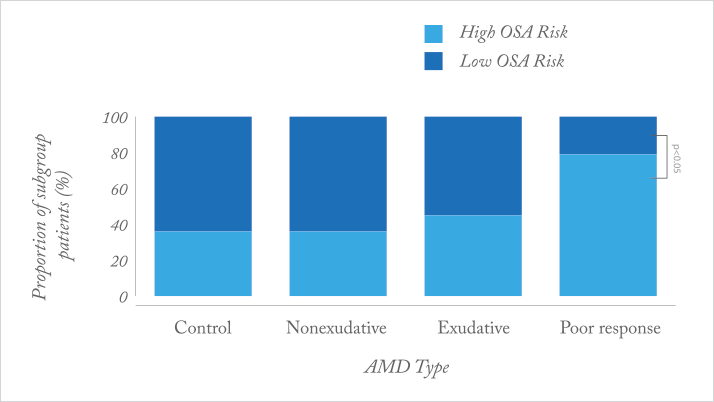
Why do some patients respond to anti-VEGF therapy well, and others do not? What makes for a poor responder? It appears that between seven and 15 percent of all patents with age-related macular degeneration (AMD) or diabetic macular edema (DME) are considered to be “poor responders” to anti-VEGF agents (1–4). A recent prospective case-control study (5) identified one possible factor: obstructive sleep apnea (OSA). Patients with newly-diagnosed AMD and DME were enroled, and treated as needed with intravitreal bevacizumab. Poor responders with AMD were defined as having persistent subretinal fluid on OCT after ≥3 injections; poor response in patients with DME was defined as patients needing ≥3 consecutive injections, and patients’ risk of having OSA was assessed using the Berlin questionnaire.
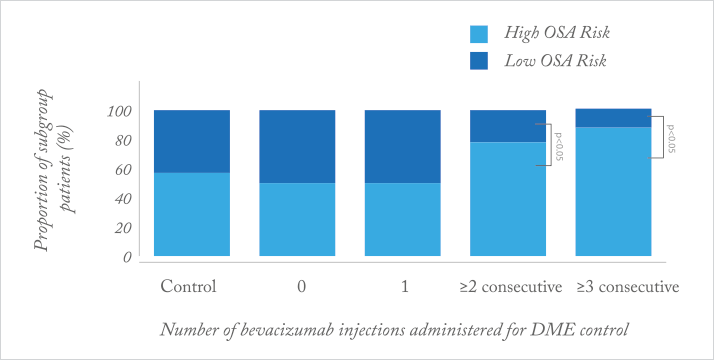
What the investigators found was that patients with exudative AMD (Figure 1) and DME (Figure 2) who are poor responders to intravitreal bevacizumab are significantly more likely to be at a high risk of OSA, relative to age-matched controls. Why? OSA causes transient hypoxia, and is associated with inflammation and vascular endothelial dysfunction – all of which can induce VEGF expression in the retina. It seems clear that there are a range of concomitant comorbidities that can affect the retinal vasculature and exacerbate or accelerate diseases like wet AMD or DME through elevated retinal VEGF expression. Accordingly, fixed doses and dosing intervals might not “cut the mustard” in such patients.
References
- PJ Rosenfeld et al., “Characteristics of patients losing vision after 2 years of monthly dosing in the phase III ranibizumab clinical trials”, Ophthalmology, 118, 523–530 (2011). PMID: 20920825 I Krebs et al., “Non-responders to treatment with antagonists of vascular endothelial growth factor in age-related macular degeneration”, Br J Ophthalmol., 97, 1443–1446 (2013). PMID: 23966368. A Lux et al., “Non-responders to bevacizumab (Avastin) therapy of choroidal neovascular lesions”, Br J Ophthalmol., 91, 1318–1322 (2007). PMID: 17537784 T Otsuji et al., “Initial non-responders to ranibizumab in the treatment of age-related macular degeneration (AMD)”, Clin Ophthalmol., 7, 1487–1490 (2013). PMID: 23901256. BLW Nesmith et al., “Poor responders to bevacizumab pharmacotherapy in age-related macular degeneration and in diabetic macular edema demonstrate increased risk for obstructive sleep apnea”, Retina, 32, 2423–2430 (2014). PMID: 25062438.
