It’s well known that age-related macular degeneration (AMD) is a multifactorial disease, with genetic factors playing a significant (but not fully understood) role. Those with a family history of AMD are at a greater risk of developing it, and developing it earlier, than those without. But how large a role do genes really play? Anneke den Hollander of the Radboud University Medical Center in Nijmegen, The Netherlands, and her colleagues explored some of the rarer genetic variants implicated in AMD (1). They performed a retrospective case-controlled study of 22 families (comprising 114 people with AMD and 60 without), and a case-control cohort of 1,589 unrelated AMD patients and 1,386 unaffected individuals enrolled in the European Genetic Database (EUGENDA). All AMD control participants provided a detailed questionnaire of their medical history, age of onset of first AMD symptoms, family AMD history, and a series of lifestyle factors. A positive family history of AMD was defined as individuals with a minimum of two first-degree relatives with AMD, either suspected or confirmed. The researchers then used whole-exome sequencing to analyze the genotypes of 85 affected members of families with a history of AMD.
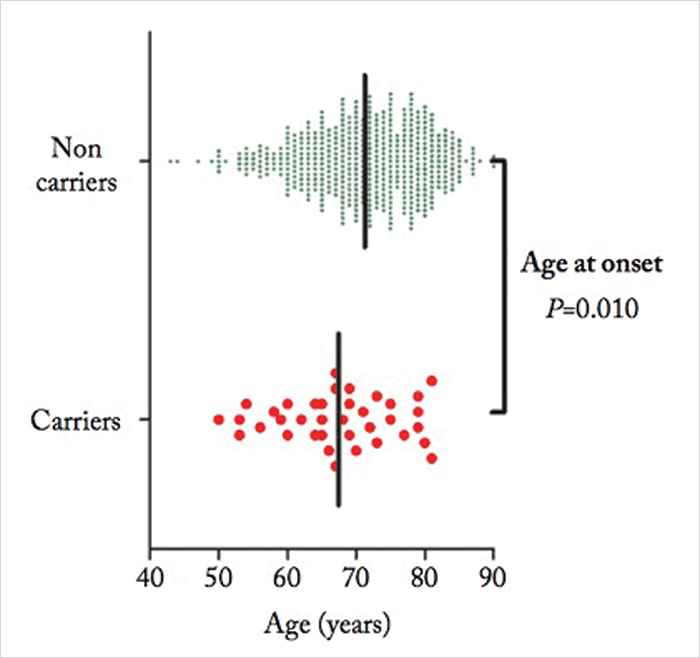
The rare variants being studied are complement pathway genes – CFI, C9 and C3, so the levels of the complement component C3 and the activation fragment C3d proteins were measured in all subjects too. Defective regulation of the alternative complement pathway is known to contribute to the development of AMD (2), and is thought to cause inflammation, immune deposits and damage to retinal pigment epithelial cells. “Many of the genetic variants associated with AMD reside in genes encoding components of the complement cascade. Also, elevated levels of complement activation have been measured in plasma samples of patients with AMD versus controls,” says den Hollander. “Patients carrying these rare variants differ clinically from non-carriers by an earlier age of onset, and they more often report a positive family history. Also, patients with advanced atrophic AMD carry these variants more frequently than patients with neovascular AMD,” she adds (see Figure 1).
Although the rare variants were found in five of the 22 families with AMD (see Table 1), they didn’t always segregate with the disease – reinforcing the point that ultimately, disease development is owing to a mix of genetic, environmental and lifestyle factors. So would it be worth developing a genetic test?
“Family members of patients with AMD often want to know their own risk,” says den Hollander, “and genetic testing could potentially be offered, alongside counselling. This might encourage people to take steps to lower their risk, such as quitting smoking, exercising more or taking antioxidant supplements.” But when many of the people carrying these variants won’t go on to develop AMD, is it justified? “In the future, genetic tests may become important if clinical trials or new treatments are based on genotype. However, as this is not yet the case, genetic testing is currently not recommended by the American Academy of Ophthalmology,” concludes den Hollander.
References
- NT Saksens et al., “Rare genetic variants associated with development of age-related macular degeneration”, JAMA Ophthalmol, [Epub ahead of print] (2016). PMID: 26767664. PF Zipler et al., “The role of complement in AMD”, Adv Exp Med Biol, 703, 9–24 (2010). PMID: 20711704.
