- Predicting which patients are at a high risk of developing post-LASIK keratectasia can be a challenge, particularly in cases of suspect and forme fruste keratoconus
- A range of parameters can be used to try to distinguish between normal and at-risk patients, but none are perfect; the challenge is finding the right combination
- Clear terminology, effective statistical analysis and a useful clinical model all help when developing an artificial intelligence tool to assess a patient’s risk
- A comprehensive statistical analysis combining a number of parameters can differentiate normal and abnormal corneas with a high rate of success
Corneal ectasia can be a disaster for patients. Blurred vision, light sensitivity, frequent changes in spectacle prescription, halo, ghosting, headaches, eye irritation… and later corneal scarring and hydrops – are all potential consequences of the progressive steepening and thinning of the cornea, and all can conspire to dramatically reduce the quality of life of these patients. Keratectasia can come in a primitive form, as we see in patients with keratoconus or pellucid marginal degeneration, or an iatrogenic form, like we see after refractive surgery on patients with undiagnosed, subclinical keratoconus. Clearly, anything that’s iatrogenic is something we need to minimize the occurrence of. So how can we detect the earliest manifestations of keratectasia so that we can avoid performing laser refractive surgery on these patients? And what terminology do we use to label these corneas? Understanding exactly how to assess and diagnose these cases is key to making the right treatment decisions. Often, this isn’t a simple process. To give us the best chance of detecting corneas that are susceptible to keratectasia, we need to do three things: clarify our terminology, develop appropriate statistical methods, and use an applicable clinical model.
What’s in a name?
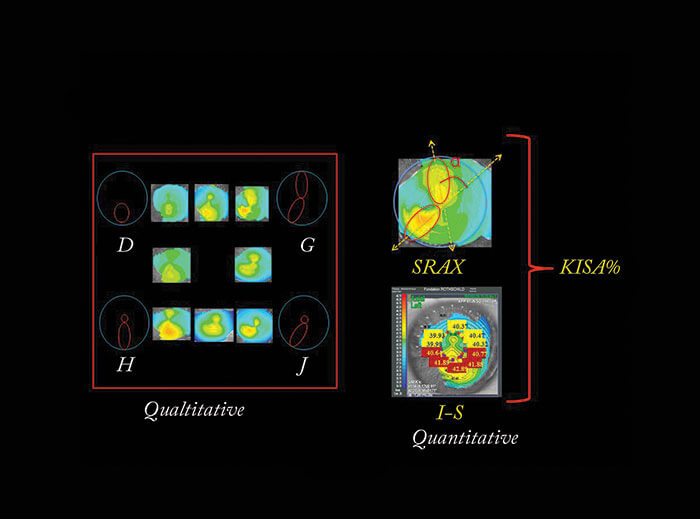
First, there’s terminology. For example, is your subclinical keratoconus better described as forme fruste, or suspect? The term “keratoconus suspect” describes corneas that show anterior topographically detectable features that are evocative of subclinical disease. These features were first described qualitatively by Rabinowitz and colleagues (1), but their description was limited by the smooth transition of topographical aspects (such as inferior steepening) from normal to suspect and subsequent keratoconus. The use of quantitative videokeratography-derived indices (2) such as the SRAX (skewed radial axis) or the I-S (difference between inferior and superior keratometry measured at 5 mm) represents a more reproducible way of quantifying keratoconus and its early phenotypes, and reduces the complexity of proper classification (Figure 1). In medicine, “forme fruste” means an incomplete, abortive, or unusual form of a disease or syndrome. Stephen Klyce (3), described forme fruste keratoconus as the fellow eye of a keratoconic cornea that has no clinical findings of any sort except subtle characteristics suggestive of an early subclinical keratoconus. Essentially, this is a very low phenotypic expression of the keratoconus that is below the threshold of Placido detection.Smart statistical methods…
With the current, gold-standard, diagnostic tools, the classification of a cornea as “normal” doesn’t exclude the possibility of subclinical keratoconus. The sensitivity of computer-assisted Placido-based videokeratotopography is not sufficient, and an I-S value above 1.4 D or a steep keratometry (>47 D) may not necessarily be an indicator of a keratoconic subtype – in other words, the specificity of Placido topography is not 100 percent. Ideally, we want a test that can perfectly separate normal and abnormal corneas – but such a perfect test doesn’t exist right now. To improve the accuracy and precision of this kind of screening, what we need to do is combine several of the corneal parameters obtained from Placido and elevation topography together into a discriminant function (4). Linear discriminant analysis is a technique that attempts to model the differences between different classes of data, by looking for linear combinations of variables that best explain the data. For example, combining I-S indices with the thinnest pachymetry measurements and posterior elevation data detects subclinical keratoconus with far greater accuracy than I-S alone.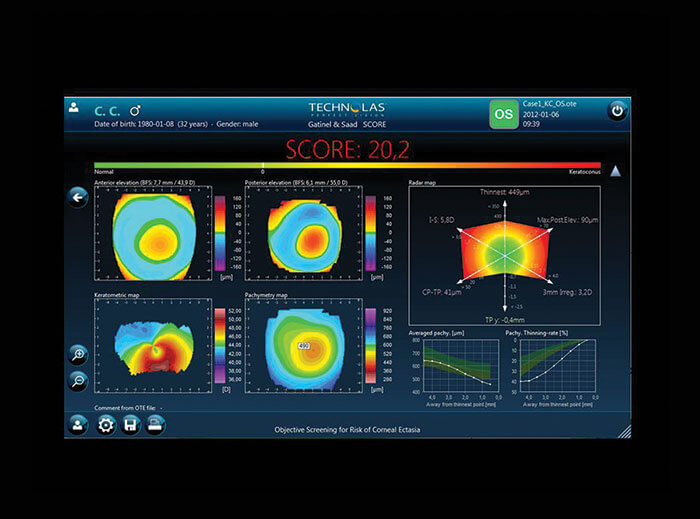
… and clever use of clinical models
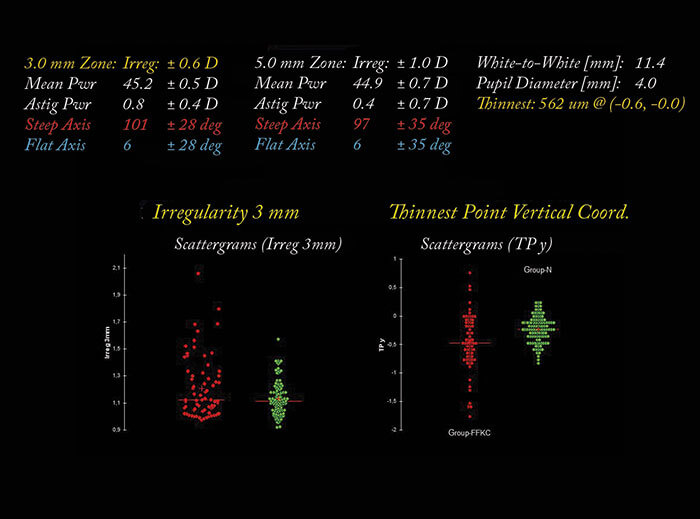
To fully evaluate and characterize such an approach, we need a pertinent clinical model. We know that keratoconus has a genetic component, and in cases of unilateral keratoconus, we know that both eyes have the same genetic makeup – and that even if one eye looks normal (based on Placido topography), it probably represents a false negative. Damien Gatinel and I studied the contralateral corneas from cases of unilateral keratoconus (4), on the basis that these eyes were giving false negative results using current diagnostic methods. First, we performed an Orbscan (Bausch+Lomb) examination for cases of “unilateral” keratoconus. Orbscan provides us with Placido topography and anterior elevation, posterior elevation and pachymetric maps information. We also selected a group of bilateral normal eyes that had LASIK and three years of follow-up, without developing ectasia and those corneas also benefits from an Orbscan examination. Then, we compared all the parameters that we had measured with the Orbscan between the normal eyes and the eyes that only appeared to be normal – i.e. those with forme fruste keratoconus. The results were interesting – some corneal indices were significantly different between the two groups. We observed differences in posterior elevation, thinnest point, decentration of the thinnest point, difference between central and thinnest pachymetry, 3 mm zone irregularity, and between inferior and superior keratometry (I-S) (Figure 2). But again, when using just one of these parameters alone – for example a 3 mm zone irregularity – it’s not possible to separate between the groups; you get a lot of overlap (Figure 2). So this is where a good statistical method like linear discriminant analysis (that combines these parameters together) can be a big advantage.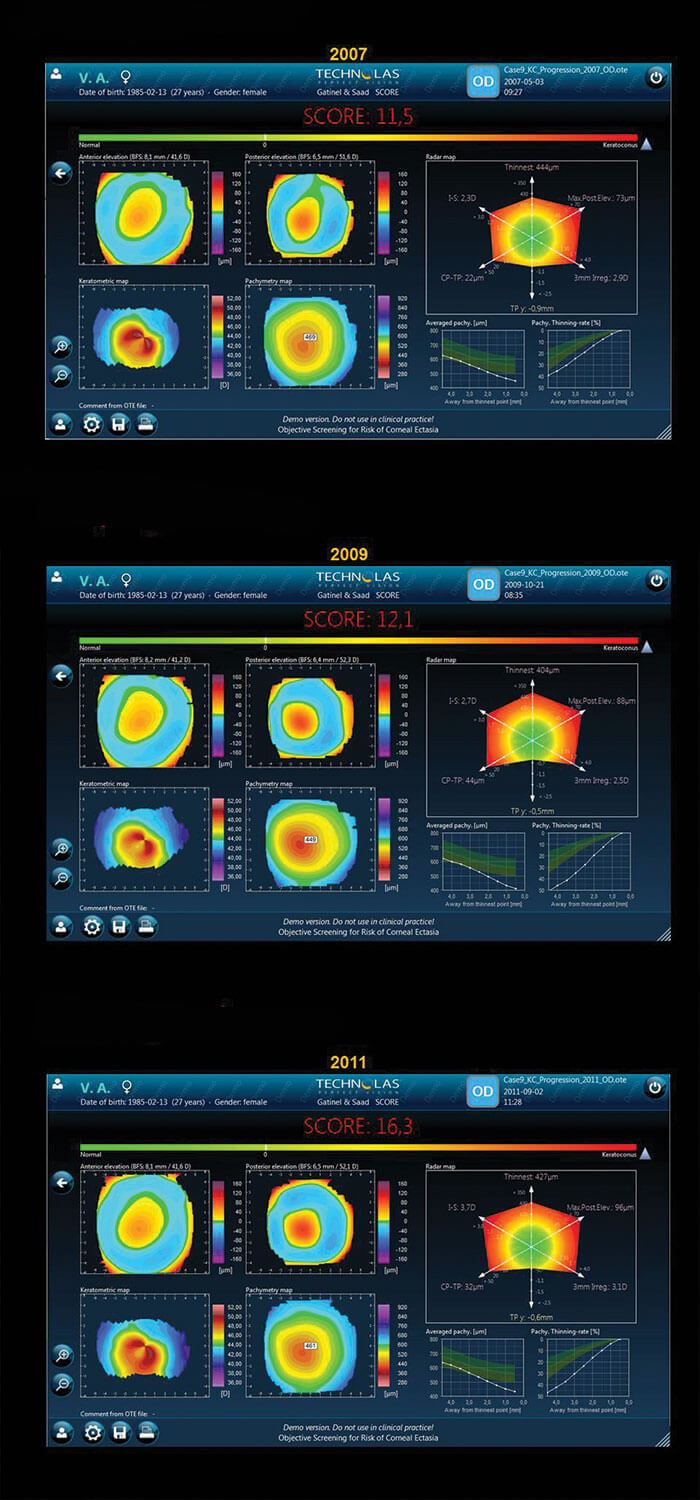
Combination is key
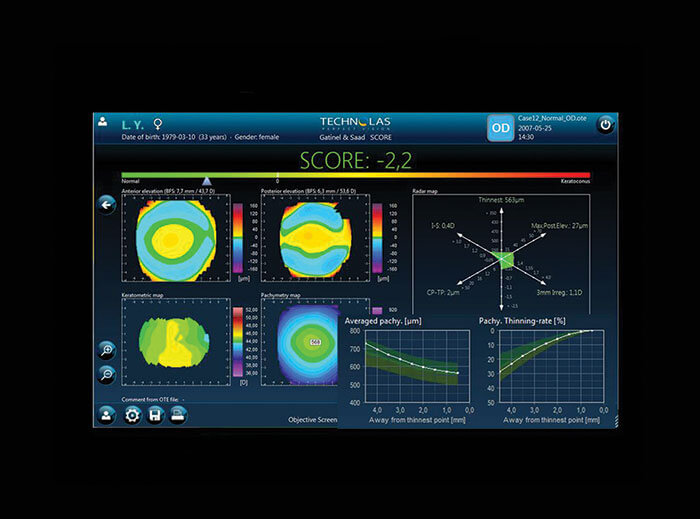
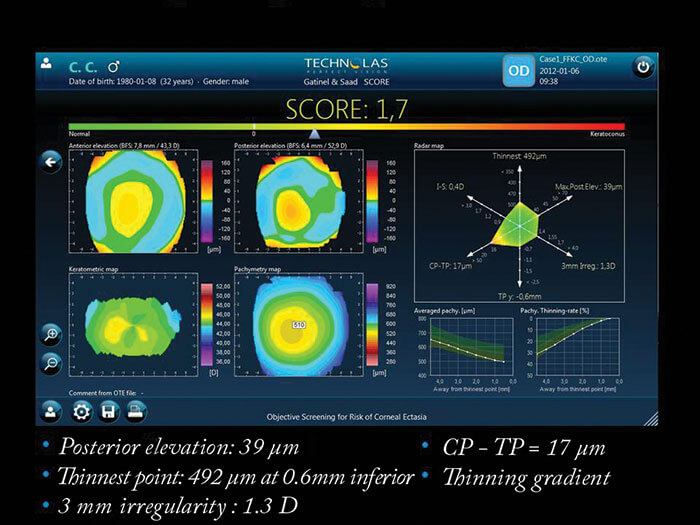
So it’s clear that combining our parameters was the most successful way to spot suspicious corneas, and it’s possible to automate the process. We developed the SCORE (Screening for Corneal Objective Risk of Ectasia) Analyzer, that combines different significant parameters for the detection of early keratoconus into a single number and this artificial intelligence system can now be used on the Orbscan II system. Figure 3 shows a normal cornea, with a SCORE of -2.2 – the most important parameters are displayed on a color scale map (Radar), which includes the thinnest point, the difference between central pachymetry and thinnest pachymetry, the posterior elevation, the 3 mm irregularity, I-S and the decentration of the thinnest point. Based on the color map, you can see that green areas are normal, yellow is suspicious, and red is abnormal. A forme fruste keratoconus is shown in Figure 4, so here the SCORE is slightly positive at + 1.7. In the radar map, there is green but also some yellow, and the pachymetry thinning rate (as first described by Renato Ambrósio et al (5) is at the limit of the normal range. You don’t really need to use the SCORE analyzer to detect advanced keratoconus (Figure 5), but it can be used to objectively evaluate keratoconus progression, as is shown clearly in Figure 6: in 2007 (a) the patient’s score was 11.5, in 2009 (b) it was 12.1, and in 2011 (c) it had increased to 16.3. The same approach can be generalized to other devices – we are currently investigating an artificial intelligence system that uses optical path difference (OPD) scans that combine higher order aberration detection with Placido indices in order to obtain an overall corneal score. Trying to distinguish normal from early pathological cases is a common challenge in medicine (6), and diseases that are multifactorial are the most challenging of all in terms of designing appropriately sensitive and specific tests. Techniques such as linear discriminant analysis (determining a linear combination of features that characterize or separates two or more disease states) may help to increase the efficiency of screening tests for early subclinical keratoconus identification – helping avoid iatrogenic keratoconus, and the disastrous consequences it can have in patients.References
- YS Rabinowitz, PJ McDonnell, “Computer-assisted corneal topography in keratoconus”, Refract Corneal Surg, 5, 400–408 (1989). PMID: 2488838. YS Rabinowitz, K Rasheed, “KISA% index: a quantitative videokeratography algorithm embodying minimal topographic criteria for diagnosing keratoconus”, J Cataract Refract Surg, 25, 1327–1335 (1999). PMID: 10511930. SD Klyce, “Chasing the suspect: keratoconus”, Br J Ophthalmol, 93, 845–847 (2009). PMID: 19553507. A Saad, D Gatinel , “Topographic and tomographic properties of forme fruste keratoconus corneas”, Invest Ophthalmol Vis Sci, 51, 5546–5555 (2010). PMID: 20554609. R Ambrósio et al., “Corneal-thickness spatial profile and corneal-volume distribution: tomographic indices to detect keratoconus”, J Cataract Refract Surg, 32, 1851–1859 (2006). PMID: 17081868. D Gatinel, A Saad, “The challenges of the detection of subclinical keratoconus at it earliest stage”, Int J Keratoco Ectatic Corneal Dis, 1, 36–43 2012).
