
It’s clear that MIGS approaches offer many advantages; in addition to offering effective IOP control and rapid visual rehabilitation, many are ab interno techniques, which reduce trauma and spare the conjunctiva. Plus, they are compatible with earlier surgical intervention. Nevertheless, not all MIGS-receiving patients reach their target IOP, and in these cases, the norm is to add topical eyedrops into the mix – the so-called MIGS ‘n’ Meds approach – but this isn’t ideal. Eyedrops can cause local side-effects (1)(2), they are difficult for elderly patients to use (3) – and then there’s the issue of cost, so it’s considered a “win” when taking a MIGS approach reduces the number of eyedrops a patient needs for IOP control. Almost inevitably, topical drop regimen adherence levels are poor: ~51–56 percent (4), or lower (5). In terms of side-effects, one of the biggest problems is corneal epithelial cell toxicity, ocular surface disease and periorbitopathy (2)(6)(7)(8)(9)(10). This downside to eyedrops is obviously unsatisfactory – after all, the objective is to ameliorate the effects of glaucoma, not to subject the patient to a suite of iatrogenic conditions. Isn’t there a better way?
Enter SLT
Selective laser trabeculoplasty (SLT) was introduced in 2001 as a replacement for argon laser trabeculoplasty (ALT). It rapidly gained acceptance as an effective therapeutic modality that’s applicable to all stages of glaucoma treatment. Recent years, however, have seen a decline in SLT use – partly as a consequence of the rising popularity of MIGS. But we have found SLT and MIGS are not mutually exclusive, but entirely complementary – synergistic, in fact. Firstly, the diagnostic value of SLT is often under-appreciated. Today, we do not have a good non-invasive diagnostic technology to help us determine the site of resistance in the natural outflow system in patients with increased IOP. Current imaging technologies do not have the ability to provide us this information. But SLT may serve as a surrogate. Since SLT primarily addresses the trabecular meshwork (TM), a successful SLT procedure implies that the primary blockage was located in the TM. Conversely, a less successful outcome post-SLT would indicate that the blockage may reside in some other part of the outflow system, such as Schlemm’s canal or the collector channels (Box 1). So, in cases where SLT is effective, a surgeon might conclude that future treatments could comprise devices that target only the TM (such as the iStent), while a poor post-SLT outcome might prompt a surgeon to opt for other procedures, such as ab interno canaloplasty (ABiC), which open up both Schlemm’s canal and the collector channels. In other words, SLT is the first step in helping to determine the health of the patient’s outflow system, and hence what treatment options are most suitable. It is important to note SLT is a non-destructive procedure, unlike ALT, and does not inhibit the use of any of the MIGS devices. Of course, SLT is far more than just a diagnostic modality – there are good reasons why this procedure replaced ALT in clinical practice. In open-angle glaucoma (OAG) and ocular hypertension, published work has shown that SLT is of equivalent efficacy to one or more medications. For example, one prospective, non-randomized study that involved 45 eyes with primary OAG (POAG) and ocular hypertension showed that SLT reduced IOP by 30 percent (~7.7 mmHg) in eyes that had not received prior treatment (11). Furthermore, this level of improvement is maintained over time: a separate study showed IOP reductions of 24.3 percent, 27.8 percent, 24.5 percent and 29.3 percent at years 1, 2, 3 and 4, respectively (12). Unsurprisingly, a systematic meta-analysis of one-year efficacy data indicated that SLT was comparable to medication with prostaglandin analogues (13). Comparing the above findings with the poor one-year adherence associated with eye-drops – and bearing in mind the significant, long-term side-effects commonly associated with these medications – it is difficult to understand why SLT is not more frequently used as an alternative to medication. Certainly, there are no major concerns with the safety of the procedure (other than a minority of SLT-receiving patients might experience transient, self-resolving IOP spikes and inflammation) and the most common adverse effects are limited to very mild discomfort for one or two days after the procedure. The consensus is that the risk-benefit ratio of SLT is highly attractive (14).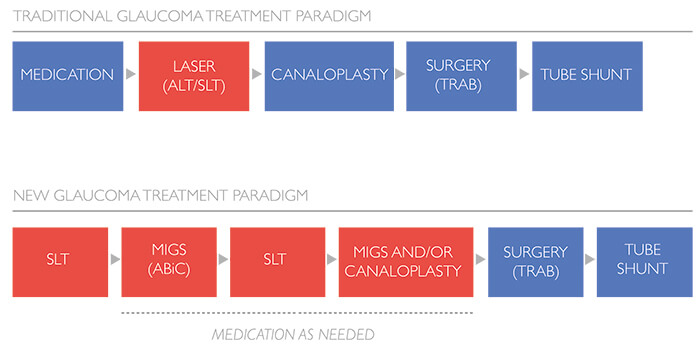
By Inder Paul Singh I think SLT is an under-used modality. It has served me well, both before and after MIGS. A retrospective analysis of our iStent data (one-year follow-up, in patients who did or did not respond well to SLT) showed that the good SLT responders had better outcomes after the iStent than the poor SLT responders. To me, this suggests that SLT may be a pre-operative indicator of the site of outflow resistance. The reasoning is that SLT works primarily on the TM; therefore, if SLT works well, it’s likely that the TM is a major part of the resistance pathway, and I might perform a trabecular bypass procedure only. And if SLT is ineffective, I’m inclined to suspect that there is a site of resistance is in the distal Schlemm’s canal; under those circumstances, I would not likely perform only a TM-directed procedure. So, as a pre-operative measure, I believe SLT has multiple benefits: not only does it result in a reduced medication burden, but also it helps the surgeon diagnose the nature of the drainage problem and adapt his or her MIGS strategy accordingly.
True synergy: SLT ‘n’ MIGS, MIGS ‘n’ SLT
Perhaps most exciting of all is the complementarity of SLT and MIGS, which, as noted, are not ‘either/or’ approaches. The diagnostic utility of SLT, in terms of guiding subsequent MIGS procedures, is now well-known. But the procedure also has immense potential for application after MIGS procedures have been carried out – it can still lower IOP in post-MIGS-procedure. SLT has the potential to entirely disrupt the current MIGS ‘n’ Meds paradigm, and begs the question: why prescribe eye-drops for patients (and have them suffer all the consequences) when SLT is just as efficacious and free of significant side-effects? The answer to the question would leave many finding it difficult to justify not using SLT alongside MIGS. Moreover, in addition to its proven safety and efficacy, the procedure is simple enough to be carried out in-office. What reasons could clinicians have to opt for medication instead of SLT? Paradigms are often hard to change. Be that as it may, the benefits of the SLT-MIGS combination are now recognized by many practitioners. Steven Vold reports that a supplemental SLT can lower IOP by an additional 2–3 mmHg and thus achieve pressures in the mid-teens: “That can mean your patient does not have to return to medications,” he says. Such a result is the consequence of true synergy between SLT and MIGS. ABiC seems to be particularly compatible with SLT: a recent study (15) reported that SLT can actually contribute to dilation of Schlemm’s canal – perhaps simply as a consequence of increased trabecular outflow – thereby contributing to additional IOP reduction. At the same time, ABiC not only clears the Schlemm’s canal and the collector channels, but also contributes to improving the function of the TM itself, by clearing TM herniations from the collector channels and separating the compressed tissue planes of the TM.Putting the patient first
SLT and MIGS both complement and augment each another – particularly in the case of MIGS procedures, such as ABiC, which open up Schlemm’s canal and the collector channels. Given that both ABiC and SLT restore and rejuvenate the outflow system, some degree of synergy would be expected. In addition, the diagnostic advantages of SLT should not be overlooked; if SLT determines that the TM is not the primary site of pathology, it is logical to proceed to the ABiC/ iTrack system, which addresses the Schlemm’s canal and collector channels in addition to TM. In the end, of course, the most important consideration is the effect on the patient; given that SLT-ABiC synergies may make the difference between a patient suffering the burden of eye-drops and being entirely medication-free, this combination approach surely deserves the attention that it is increasingly receiving.
References
- R Meda et al., Br J Ophthalmol, 101, 120–125 (2017). A Robciuc et al., Cornea, 36, 1249–1255 (2017). R Gupta et al., J. Glaucoma, 21, 189–192 (2012). R Sheer et al., J Manag Care Spec Pharm, 22, 808–817 (2016). A Joseph and LR Pasquale, Seminars in Ophthalmology, 32, 86–90 (2017). C Baudouin et al., Invest Ophthalmol Vis Sci, 48, 4123–4128 (2007). C Baudouin et al., Ophthalmology, 115, 109–115 (2008). N Jaenen et al., Eur J Ophthalmol, 17, 341–329 (2007). F Pérez-Bartolomé et al., Eur J Ophthalmol, [Epub ahead of print] (2017). C Patradul et al., Asia Pac J Ophthalmol, 6, 238–242 (2017). S Melamed et al., Arch Ophthalmol, 121, 957–960 (2003). LJ Katz et al., J Glaucoma, 21, 460–468 (2012). AL Freitas et al., Arq Bras Oftalmol, 79, 417–427 (2016). Y Barkana and M Belkin, Surv Ophthalmol, 52, 634–654 (2007). A Skaat et al., J Glaucoma, 26, 361–366 (2017).
