In the last couple of decades, Photobiomodulation (PBM) therapy – a light-based treatment approach used widely for tissue repair and reducing pain and inflammation – has gained recognition among biomedical researchers and healthcare professionals. Now, LumiThera’s Valeda Light Delivery System is using a multi-wavelength PBM technology to treat ocular diseases and damage, aiming to help physicians address unmet patient needs.
Janis Eells, Professor of Biomedical Sciences, University of Wisconsin-Milwaukee, WI, USA, has spent over 20 years researching the use of PBM in preclinical ophthalmology indications.
Tell us about PBM.
I’m a pharmacologist, and my expertise lies in visual toxicity and retinal disease. I became interested in the role that mitochondria play in these processes – they are energy producers and master regulators of our metabolism. I researched animal models of toxicity-induced blindness for years, and became involved in investigating the healing benefits of using PBM in ocular diseases connected to mitochondrial dysfunction in aging, toxic injury and diseases including retinitis pigmentosa, diabetic retinopathy (DR), and age-related macular degeneration (AMD).
PBM works by activating mitochondrial respiratory chain components and stabilizing their metabolic function. It is applicable to a broad spectrum of retinal and optic nerve disorders – from inherited retinal disorders, AMD, diabetic eye diseases, and glaucoma, to trauma and wound healing. Current treatments for ocular disease work on symptoms, rather than targeting the underlying pathology of the disease. In retinal disease and/or damage, there is an increase in mitochondrial dysfunction and oxidative damage. The effects of PBM at the cellular level include increasing ATP generation, modulation of intracellular signaling molecules, including reactiveoxygen species (ROS) and nitric oxide (NO) and secondary effects that culminate in sustained changes in cell function and viability. Improvement in these cellular outcomes leads to benefits at the clinical level.
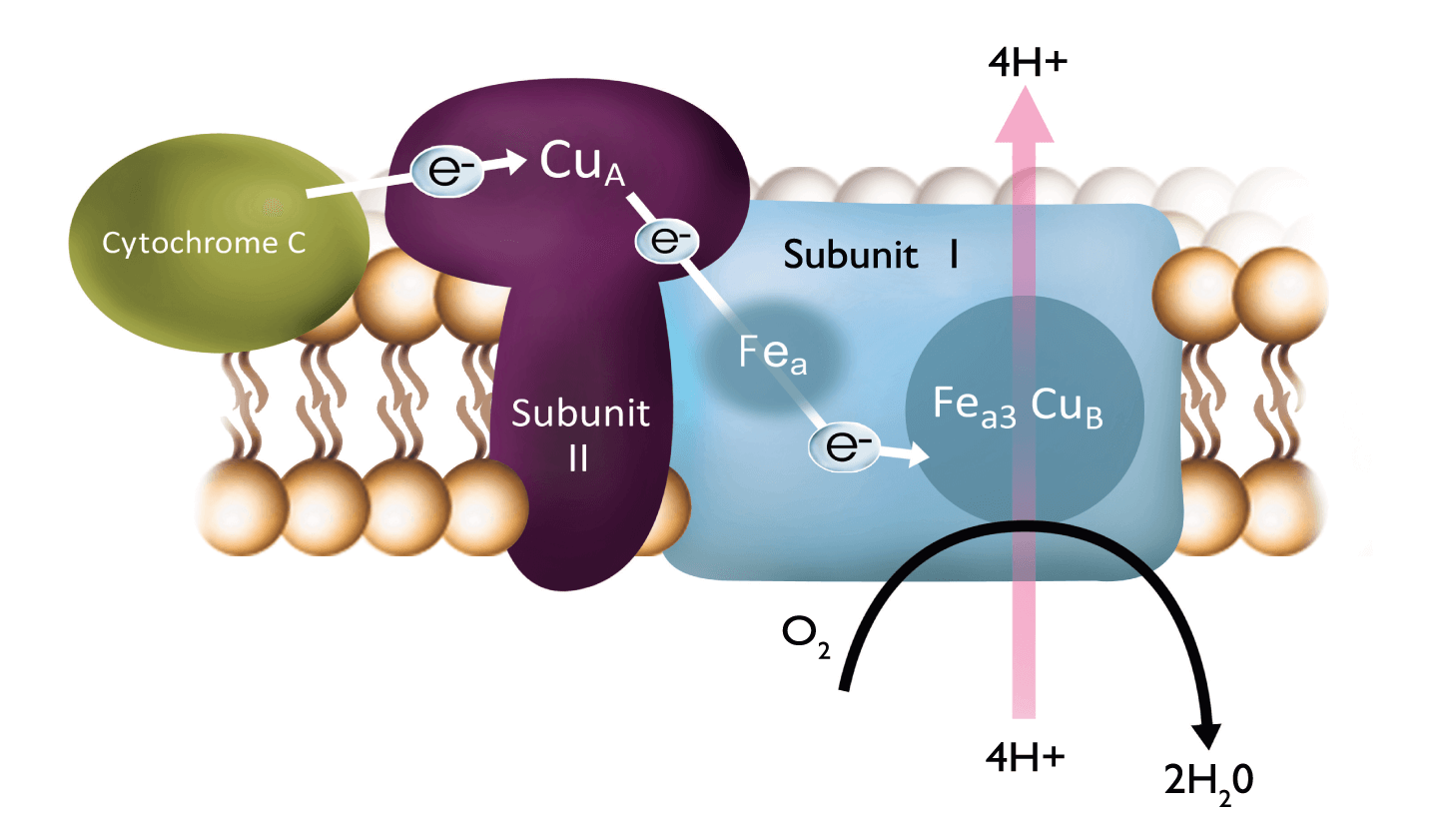
The photobiomodulatory effects of PBM at the molecular level begin with the absorption of specific wavelengths of light by a key mitochondrial enzyme, cytochrome c oxidase (CcO). The interaction of light with CcO creates a biochemical response. Just as the chlorophyll molecules in plants absorb light and turn it into energy stimulating growth, the mitochondria in animal cells can use light to stimulate cell function and activate protective intracellular pathways. The process only works when the mitochondria are not functioning properly – if the cells is healthy, the light will not improve its function, and no beneficial effect is observed. In a malfunctioning cell, mitochondrial output becomes impaired and energy production can become reduced resulting in cellular dysfunction and subsequent clinical complications. Treatment with PBM can stimulate CcO to optimize its output and thereby improve mitochondrial function. PBM also activates transcription factors in the nucleus resulting in changes at the molecular level which aid in cellular response and resets the metabolic environment in the cell.
It’s important to note that the dose or fluence has to be just right; too little light will not have the desired effect, and if there is too much, the beneficial effect is turned off. This “just right” does can be very difficult to determine in the case of more traditional pharmacological treatments. Research efforts in the PBM field will allow
for further optimization of light dosing to treat different indications.
Many applications of PBM, from heart disease, diabetic wound healing, skeletal muscle injuries, spinal cord healing to neurodegenerative diseases such as Alzheimer’s, traumatic brain injuries, and many more are being investigated. PBM has a tremendous potential in preventive medicine as well.
Marion Munk, ophthalmologist at Inselspital University Clinic Bern in Switzerland, who specializes in researching retinal imaging in the area of retinal and inflammatory diseases, talks about her experience with PBM for treating dry AMD.
Dry AMD currently has no recognized treatment. How can PBM change this?
I was shown early on images of patients with dry AMD that were treated with PBM and they showed significant reductions in drusen. Our imaging center was enlisted to conduct a masked review of the optical coherence tomography (OCTs) generated during the PBM clinical trials. Usually in the progression of AMD, we see a slow increase in drusen volume – and in the area affected by it. If the patient has advanced dry AMD, we see progression in lesion growth of geographic atrophy areas. In some patients, there is a reduction in drusen volume right before the development of advanced dry AMD; which is characterized by the development of atrophy of the outer retina and retinal pigment epithelium. Patients treated with PBM show a decrease of drusen volume over the treatment time – and this is not linked to an onset of geographic atrophy or any precursors.
Tell us about your involvement in the LIGHTSITE I clinical trial…
The LIGHTSITE I trial evaluated the effect of PBM as a treatment for dry AMD, using LumiThera’s Valeda Light Delivery System. In this first double masked trial investigating efficacy and safety of PBM for this indication, patients underwent two series of treatments, three times per week for three to four weeks, with a repeated treatment series conducted after six months. Outcome measures included best-corrected visual acuity (BCVA), contrast sensitivity, microperimetry, central drusen volume, drusen thickness, and a quality of life assessment.
PBM-treated subjects show improvements in BCVA with approximately 50 percent of subjects showing more than five letter gains compared with sham controls. Subjects also showed improvements in contrast sensitivity and quality of life assessments. Subjects treated with PBM show a reduction in drusen volume following PBM. Not surprisingly, patients who showed less advanced stages of AMD benefited most.
At the moment, the LIGHTSITE II trial is taking place across Europe, in conjunction with the LIGHTSITE III study trial, which is being conducted at multiple centers in the USA. I have found the data of the LIGHTSITE I trial to be very encouraging, and we now need the multi-center trials data to confirm our previous observations.
How could PBM ultimately make an impact on the outcomes of dry AMD patients?
If the results of the multi-center trial are positive, it will have a huge impact on patients with dry AMD. For the first time we will have a treatment available for this patient cohort beside nutritional supplements. The next Holy Grail question is whether PBM will reduce the progression of the disease, which is of uppermost importance. We live in very exciting times!
Clark E. Tedford, CEO and President of LumiThera, explains the science behind the multiwavelength PBM approach
One of the important aspects of PBM is that different wavelengths produce different effects. When designing Valeda, we had to choose the right targets and select the appropriate PBM wavelengths – from the visible to near infrared light.
The enzyme Cytochrome c Oxidase (CcO) absorbs light at two different wavelengths, which are critical for the optimal PBM functioning by the Valeda system. These wavelengths are 660 nm – far red – and 850 nm – infrared. We could have developed a system that would use only one of those wavelengths, but when assessing the targets, we found that they were both critical for the enzyme to work. Each wavelength targets a different, but critical site of CcO function. Next, we decided to add a third wavelength based on different literature publications. We knew that the disease progresses to Geographic Atrophy and 10-15 percent of dry AMD patients’ disease convert to wet AMD. In the literature, the 590 nm wavelength – yellow – improves nitric oxide generation in ischemia conditions potentially improving local blood flow, and we also saw that it inhibits VEGF expression in RPE cells. We felt that we might be able to slow disease progression and reduce the number of patients converting to wet AMD, and/or allow us to treat wet AMD patients. Given that each one of these wavelengths had specific and unique targets, by putting them together we knew we could get the most benefit out of the Valeda system.
We created a delivery system in Valeda that allows us to optimize each wavelength individually and provides us with an adaptable therapy. We are now beginning to investigate the use of Valeda in diabetic macular edema and other ocular areas. Each disease may require further study, but we have a good starting place from our studies in dry AMD. It is an evolving project; our ultimate aim is to create a system that can be optimized for different treatments. We believe the use of PBM in ophthalmology is just beginning. We are excited to get the PBM technology platform into the hands of physicians to work with us and provide new treatments for their patients.
