
When managing a patient with primary open angle glaucoma (POAG) or ocular hypertension (OHT), standard practice is to reach for topical medications that lower IOP – eye drops. Selective laser trabeculoplasty (SLT) mediates equivalent IOP reduction to the eye-drop approach, but has rarely been used as first-line therapy. Part of the reason for this has been the lack of carefully designed head-to-head comparisons of SLT and eye-drops. A recent study (1) filled this data gap, with significant implications for clinical practice in POAG and OHT.
Status quo ante
How were the SLT and eye-drop approaches perceived prior to this trial? Let’s recap some of their key features. The toxic effects of eye-drops – particularly as a consequence of formulation preservatives, such as benzalkonium chloride – were well known; consequences include poor therapy adherence and disruption of the ocular surface to the extent that future surgery may be compromised (2). This in turn led to the advent of preservative-free topical therapies – which are relatively expensive. Consequently, the financial burden associated with glaucoma medication has risen markedly: annual costs range from ~£100 per year (for example, Monopost) to over £350 per year (Cosopt prescription free £28.59 x 12 months. Furthermore, preservative-free drops are not completely free of ocular and systemic side effects.
SLT is less well-understood than medical therapy: its exact mechanism of action remains mysterious. Studies confirm that an increase in outflow occurs, likely via multiple mechanisms after the laser stimulus. Selective Laser Trabeculoplasty (“plasty:” shape altering) suggests that structural change predominates, but studies fail to show any lasting trabecular architectural response to SLT, even with high-resolution electron microscopy. Key components of SLT’s action are likely to include recruitment of monocytes and macrophages that ‘clean out’ the trabecular meshwork and thereby improve its function (3). This outflow modulation can be very effective: SLT has been shown to mediate IOP reductions of 30-40 percent from baseline, which are then sustained for significant periods – from 30 months (4) to five years (5). Furthermore, though the effect is not permanent, the procedure can be repeated if necessary. Finally, it’s worth noting that SLT is a painless, safe outpatient procedure, often taking less than 5 minutes per eye with fast recovery times.
Status quo post
Without hard evidence of the comparative utility and efficacy of SLT and eye-drops in a real-world setting, it was impossible for clinicians to gauge their relative value. In this context, Gus Gazzard’s study in the Lancet (1) is a game-changer; indeed, it is already altering patterns of clinical practice, both within the UK and internationally. This trial (see sidebar “Casting LIGHT on First-Line SLT” and Gus Gazzard's piece here) showed that first-line SLT is at least equivalent to eye-drop medication in terms of clinical outcomes and health related quality of life, and is more cost effective. In particular, SLT was associated with better IOP control over the 36 month study period; less intense eye-drop therapy (74.2 percent of SLT patients required no eye-drops to maintain target IOP, whereas 65 percent of eye-drop group needed one medication and 35 percent required two or more medications); less disease progression; and – strikingly – zero glaucoma surgeries. Notably, within the SLT group, only one IOP spike was reported from over 700 treated eyes which is less frequent than previously thought and further highlights the very low risk profile
Some of these effects may be related to reduced diurnal IOP variation after SLT compared to dose-dependent IOP reduction with eye drop administration. Whatever the mechanism, the economic consequences are clear – for every patient given SLT first instead of eye drops, the resulting cost-savings are greater than the cost of SLT for two additional patients. Furthermore, in addition to sparing NHS resources, first-line SLT could allow nearly 75 percent of patients to avoid eye drops – and all their attendant side effects – for at least 36 months.
Practical consequences
The cost-effectiveness of first-line SLT hasprofound implications for NHS glaucoma expenditure. Consider the costs associated with preservative-free eye-drops over a three-year period – and the savings that could be realized with three years of IOP control from a single SLT procedure. First-line SLT could save ~£1,000 per patient over three years; given the numbers of glaucoma patients in the NHS, this represents a very welcome economy.
Note that additional cost-savings may be possible: for example, the finding that only one IOP spike was observed in 776 treated eyes suggests that we may be able to dispense with IOP monitoring at one hour post-laser. Similarly, the two-week IOP assessment may be redundant, as the study found that none of the 776 eyes had their management changed by observations at this time point.
In summary, first-line SLT is associated with: IOP reductions that are at least equivalent to eye-drops; reduced rates of diseases progression; lower frequency of invasive surgery; and lower cataract incidence. The problem of adherence to daily administrations of eye-drops disappears for three or more years, as does the issue of topical therapy side-effects. Unsurprising then that LiGHT is bringing first-line POAG/OHT therapy out of the dark ages!
Casting LIGHT on First-Line SLT
A multi-center, randomized, controlled comparison of SLT (followed by eye drops where necessary) and eye-drops alone in first-line treatment of POAG and OHT (ISRCTN 32038223) at six UK hospitals (1).
Population:
- 718 treatment-naive patients with POAG or ocular hypertension, 18 years or older
Methods:
- Subjects randomly allocated to SLT (35) or eyedrops (362); monitored for 36 months after treatment
- SLT: energy delivered to 360 degrees of trabecular meshwork, by 25 non-overlapping shots per quadrant
- Eye-drops: prostaglandin analogue (first line); beta blocker (second-line); carbonic anhydrase inhibitor or alpha agonist (third or fourth line)
- Primary outcome: health related quality of life (QoL) at 36 months
- Secondary outcomes included: cost-effectiveness, safety, clinical effectiveness
Results:
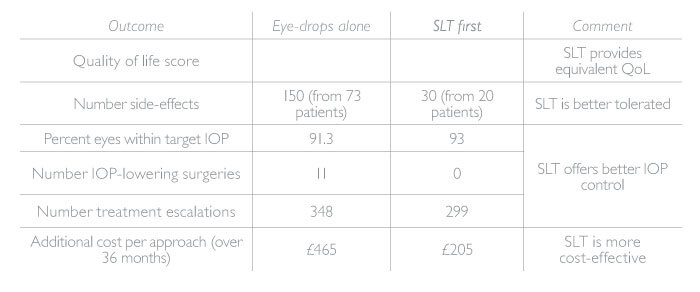
- SLT offers equivalent QoL to eye-drops, but is better tolerated and provides better IOP control (Table 1)
- Crucially, SLT is more cost effective over 36 months
Conclusions:
- SLT should be offered as standard first-line therapy for POAG and OHT.
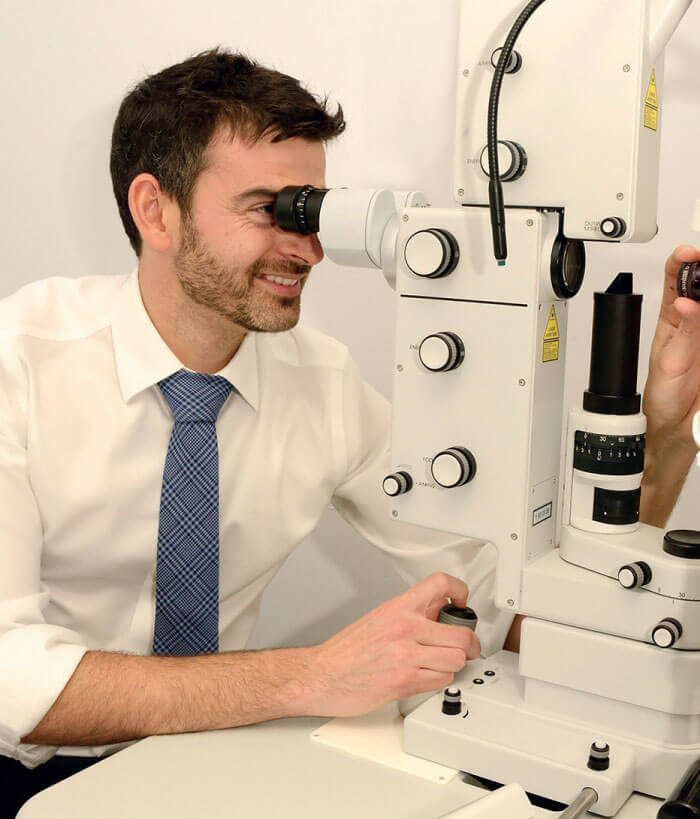
Dan Lindfield uses Optotek´s combination device OptoYag&SLT M in his clinical practice for primary SLT & adjunctive SLT to remove poorly tolerated topical medications or for additional IOP reduction.
References
- G Gazzard et al., “Selective laser trabeculoplasty versus eye drops for first-line treatment of ocular hypertension and glaucoma (LiGHT): a multicentre randomised controlled trial”, Lancet, 393, 1505 (2019). PMID: 31264958.
- D Broadway et al., “Adverse effects of topical anti-glaucoma medication II: The outcome of filtration surgery”, Arch Ophthalmol, 112, 1446 (1994). PMID: 7980134.
- J Alvarado et al., “Monocyte modulation of aqueous outflow and recruitment to the trabecular meshwork following selective laser trabeculoplasty”, Arch Ophthalmol, 128, 731 (2010). PMID: 20547951.
- T Shazly, J Smith, M Latina, “Long-term safety and efficacy of selective laser trabeculoplasty as primary therapy for the treatment of pseudoexfoliation glaucoma compared with primary open-angle glaucoma”, Clin Ophthalmol, 5, 5 (2010). PMID: 21311650.
- J Lai et al., “Five-year follow up of selective laser trabeculoplasty in Chinese eyes”, Clin Exp Ophthalmol, 32, 368 (2004). PMID: 15281969.
- Cost Comparison Charts, Regional Drug and Therapeutics Center Newcastle (2019). Available at: https://bit.ly/2FXIcBh. Accessed July 8, 2019.
