- Ocular surface disease has traditionally been treated with artificial tears and topical cyclosporine A or corticosteroids
- Such topical medications comes with the specters of poor treatment compliance and tolerability issues, as well as difficulties with effectively delivering medication to the ocular surface
- In recent years, ocular surface disease and dry eye have become increasingly common complaints at ophthalmic practices
- New treatments and technologies are evolving to redefine the eyedrop.
Ocular surface disease (OSD) is extremely common – and though it is not always the primary complaint, it is implicated in many cases that present to ophthalmic practices. Managing OSD can be challenging because the signs and symptoms are variable and often poorly correlated, and the etiology is diverse.
For many years, the mainstay of OSD treatment has been topical eyedrops, beginning with artificial tears and for more severe cases, topical cyclosporine A or corticosteroids. In recent years, we have seen significant advances in diagnostic technologies, with new point-of-service tests to measure tear osmolarity and inflammatory markers, and we can now perform meibomian gland imaging with dedicated instruments. But new treatment options have been slow to emerge. Recently, lifitegrast (Xiidra, Shire Ophthalmics) was approved in the US to treat the signs and symptoms of dry eye. Lifitegrast is an integrin antagonist that binds to lymphocyte function-associated antigen 1 (LFA-1) on T-cells to block the interaction of LFA-1 with intercellular adhesion molecule 1 (ICAM-1) – a cell surface protein that is overexpressed in patients with dry eye disease. In so doing, it is believed to inhibit T-cell activation and migration, and stop or reduce the secretion of inflammatory cytokines. Clinical studies have shown that its onset of action is less than two weeks (1). However, addressing inflammation doesn’t necessarily solve the entire spectrum of symptoms associated with OSD. The hunt for other adjunctive therapies continues…
Challenges of topical therapy
One of the reasons that we have seen so few new agents for OSD is the pharmacological challenges associated with drops for the ocular surface. Certainly, the cornea is easily accessible, but designing a drop that stays in contact with the cornea long enough to have an effect – while not causing discomfort, toxicity, or visual disturbances in the process – has been challenging. A standard eyedrop contains about 40–50µL of fluid, which is sufficient volume to activate a blink reflex that rapidly clears the drop from the surface within 15–30 seconds of instillation, through spillover and drainage through the lacrimal system (2). It is estimated that only 1–7 percent of an eyedrop’s medication has a therapeutic effect on the target tissue (3). Gels and ointments might have a longer dwell time on the cornea, but because they create an uneven refractive surface they also significantly distort vision, making their use during waking hours limited.Another challenge with eyedrops is that many compounds, including cyclosporine, are poorly soluble. Current formulations of topical cyclosporine get around this problem by suspending the drug in an oil-based emulsion, but the patient has to shake the drops before use, and multiple excipients are needed to buffer the drug and maintain suspension, with each additional excipient having the potential to affect tolerability or efficacy. Finally, even if a new topical medication can overcome the hurdles of solubility and efficacy, we know that patient compliance with eyedrops is relatively poor, even when they are prescribed an agent to treat a sight-threatening disease like glaucoma (4). In many cases, blurred vision, stinging, and other tolerability issues contribute to poor adherence.
New approaches
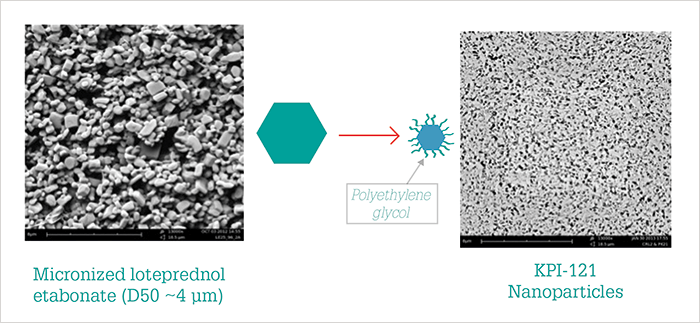
What if traditional eyedrops could be replaced with a longer lasting form of drug delivery? Sustained release implants and reservoirs have certainly been an area of intense focus in vitreoretinal therapy, but these do not easily lend themselves to corneal applications. There is a hydroxypropyl cellulose insert for sustained-release treatment of dry eye (Lacrisert, Bausch + Lomb) that has been available for many years. Certainly, some patients rely on the lubrication that this insert provides for relief, but in my opinion it is a last-resort choice because the intermittent blur caused by the sustained release of methylcellulose has a significant impact on vision. Other delivery device approaches have also been trialed. An intracanalicular depot that slowly releases dexamethasone (Dextenza, Ocular Therapeutix) recently completed a Phase III pivotal trial (NCT02736175) of its use for the control of postoperative ocular inflammation and pain after cataract surgery. As Dextenza met endpoints for decrease in inflammation, I expect that the indication for its use will expand. An iontophoresis device that delivered dexamethasone phosphate (EGP-437; EyeGate Pharma) has showed statistically significant improvements in signs and symptoms of dry eye in response to a controlled adverse environment challenge in a Phase II trial, but failed to achieve the target endpoints (corneal staining and ocular discomfort) (5). Nanoparticle technology provides a promising avenue for delivery of established drugs to the cornea and tissues beyond. Research has shown that a mucoadhesive nanoparticle drug delivery system prolongs the precorneal residence time of encapsulated cyclosporine A by adhering to mucous membranes (6). OTX-101 (Seciera) is a novel nanomicellar formulation of cyclosporine A 0.09%, acquired from Auven Therapeutics by Sun Pharma, who recently announced results from a multicenter, randomized, double masked, vehicle-controlled Phase III confirmatory study. After 12 weeks of treatment, OTX-101 showed statistically significant improvements over the vehicle control in Schirmer’s score and several key secondary endpoints (7).Similarly, Santen has a cationic nanoparticle water/oil micelle vehicle, Novasorb, which contains a positively charged surfactant and is electrostatically attracted to the negatively charged mucins on the surface of the eye, aiding retention. The vehicle is used alone for the treatment of dry eye symptoms (Cationorm), and when cyclosporin A is embedded in the oily core of the droplets (Ikervis) – although both products are currently only available in certain European and Asian markets (8)(9). Kala Pharmaceuticals is developing mucus-penetrating nanoparticle technology (KPI-121) that delivers loteprednol etabonate (an ester steroid) over approximately five days. Though topical corticosteroids have long been recognized as a highly effective treatment for the ocular surface, clinicians have been wary of their long-term use because of risks of IOP spikes and cataract development. But when loteprednol etabonate is formulated into tiny (200–400 nm) mucous-penetrating particles (Figure 1), it can get to target tissues very effectively without causing unwanted side effects; Phase II trials evaluating KPI-121 in 150 patients with a clinical diagnosis of dry eye showed significant improvements in conjunctival hyperemia and ocular discomfort at two weeks, with no difference in IOP between the drug and vehicle observed (10). Phase III trials of KPI-121 are now underway (NCT02813265).
Varying the vehicle
For perhaps the first time, we are also beginning to think of the eyedrop vehicle itself – something that was of only minor interest in the past – as a delivery system. For instance, Imprimis Pharmaceuticals are using a new technology in their LessDrops formulation that allows combination of multiple postoperative medications into a compounded single drop; active pharmaceutical ingredients that wouldn’t ordinarily mix can be solubilized into a well-distributed particle suspension. More relevant to the treatment of OSD, a non-aqueous, preservative free drug delivery system based on semifluorinated alkanes (EyeSol, Novaliq) has been shown to enhance the solubility, suspensibility and stability of drugs such as cyclosporine A, and has a well-established ocular safety profile (11)(12)(13)(14)(15). From this technology stems NovaTears, a multi-dose, preservative-free lubricating drop that has been commercially available in Europe since 2015. The low surface and interface tension from the non-aqueous drop allows it to spread quickly and uniformly over the entire ocular surface without relying on blinking for distribution; the drop volume is one-fourth that of a standard eyedrop at ~10 µL which makes it less prone to spillover. In a recent prospective, post-market multicenter study of the lubricating drop in patients with evaporative dry eye disease (n=30), significant improvements from baseline were observed at six weeks in four of five measures assessed, including a 21 point decrease in the Ocular Surface Disease Index score (13). NovaTears, with the addition of omega-3 fatty acids, is anticipated to be approved in Europe by the end of 2017. The company is also developing a cyclosporine A formulation in the EyeSol vehicle, CyclASol, and a recently completed Phase II trial in 207 patients with moderate to severe dry eye showed significant improvements in corneal staining compared with vehicle (16); pivotal trials should be initiated by 2018.The ongoing need to redefine the eyedrop
OSD is a difficult and frustrating problem to treat, yet so very prevalent in our practices. Despite the many challenges in developing new topical therapies, it is absolutely essential that we continue to evaluate new treatments that can improve patient signs and symptoms. As we continue to search and investigate new compounds, I am encouraged that new drug vehicle and drug delivery technologies offer the promise of better efficacy, less frequent dosing (and so less dependent on patient compliance), and better tolerability. Stephen Lane is Clinical Professor of Ophthalmology at the University of Minnesota and Medical Director of Associated Eye Care in Stillwater, Minnesota, USA. Financial Disclosures: Lane reports that he is Chief Medical Officer of Alcon, and is a consultant for Novaliq GmbH, Shire Ophthalmics, Ocular Therapeutix, Bausch + Lomb, and Kala Pharmaceuticals.References
- CP Semba and TR Gadek, “Development of lifitegrast: a novel T-cell inhibitor for the treatment of dry eye disease”, Clin Ophthalmol, 10, 1083–1094 (2016). PMID: 27354762. GA Peyman et al., “Perfluorocarbon liquids in ophthalmology”, Surv Ophthalmol, 39, 375–395 (1995). PMID: 7604361. D Ghate and HF Edelhauser, “Barriers to glaucoma drug delivery”, J Glaucoma, 17, 147–156 (2008). PMID: 18344762. AL Robin et al., “Adherence in glaucoma: objective measurements of once-daily and adjunctive medication use”, Am J Ophthalmol, 144, 533–540 (2007). PMID: 17686450. MA Patane et al., “Ocular iontophoresis of EGP-437 (dexamethasone phosphate) in dry eye patients: results of a randomized clinical trial”, Clin Ophthalmol, 5, 633–643 (2011). PMID: 21629568. S Liu et al., “Phenylboronic acid modified mucoadhesive nanoparticle drug carriers facilitate weekly treatment of experimentally induced dry eye syndrome”, Nano Res, 8, 621–635 (2015). Available at: bit.ly/mucoadhesive. Accessed May 18, 2017. Sun Pharma, “Sun Pharma announces positive topline results of confirmatory Phase 3 clinical trial for Seciera for treatment of dry eye”, (2017). Available at: http://bit.ly/Seciera. Accessed May 17, 2017. PY Robert et al., “Efficacy and safety of a cationic emulsion in the treatment of moderate to severe dry eye disease: a randomized controlled study”, Eur J Ophthalmol, 26, 546–555 (2016). PMID: 27515572. IKERVIS 1 mg/mL eye drops, emulsion. Summary of product characteristics. Available at: bit.ly/ikervis. Accessed May 18, 2017. Kala Pharmaceuticals, “Kala Pharmaceuticals announces positive results from Phase 2 trial of KPI-121 in dry eye disease”, (2015). Available at: http://bit.ly/KalaPharm. Accessed May 17, 2017. B Kirchhof et al., “Use of perfluorohexyloctane as a long-term internal tamponade agent in complicated retinal detachment surgery”, Am J Ophthalmol, 133, 95–101 (2002). PMID: 11755844. H Meinert and T Roy, “Semifluorinated alkanes –a new class of compounds with outstanding properties for use in ophthalmology”, Eur J Ophthalmol, 10, 189–197 (2000). PMID: 11071025. P Steven et al., “Semifluorinated alkane eyedrops for treatment of dry eye disease –a prospective, multicenter noninterventional study”, J Ocul Pharmacol Ther, 31, 498–503 (2015). PMID: 26296040. RM Dutescu et al., “Semifluorinated alkanes as a liquid drug carrier system for topical ocular drug delivery”, Eur J Pharm Biopharm, 88, 123–128 (2014). PMID: 24844949. GM Tosiet al., “F6H8 as an intraoperative tool and F6H8/silicone oil as a postoperative tamponade in inferior retinal detachment with inferior PVR”, J Ophthalmol, 956831 (2014). PMID: 24672710. Novaliq, “Novaliq announces positive topline results of Phase 2 clinical trial evaluating CyclASol in adults with moderate to severe dry eye disease”, (2017). Available at: http://bit.ly/CyclASol. Accessed May 17, 2017.
