In the developed world, contact lens misuse and corneal injury are the most common causes of corneal ulcers; approximately 60,000 new cases are seen in the US alone per year. Treatment, in principle, is simple: eradicate the cause of the ulcer, and wait for it to heal. In practice, it’s not quite that easy, as ulcers can be caused by bacteria or fungal infection, or even a mixture of the two – with these, the last thing that’s needed is the wrong initial approach. However, a range of effective (but expensive) treatment options are available, meaning that most corneal ulcers can be treated, but crucially, only if the healthcare infrastructure and financial backing are given.
Contrast this with the developing world where infectious corneal ulcers are at epidemic proportions. India, for example, sees around two million new cases per year. The causes are essentially the same as in developed countries – bacterial or fungal pathogens proliferating in the cornea following injury –often from minor injuries… but access to ophthalmic care is far worse and antibiotics are too expensive for many. Even a minor corneal ulcer can lead rapidly to severe, sight-robbing corneal infections when there is no access to eyecare. In addition, antibiotic resistance means that there is a massive global unmet need for a new and effective way of treating corneal ulcers. The developing world needs a therapeutic modality that’s not just effective, but affordable and easily used in even in the most rural of areas.
A new approach
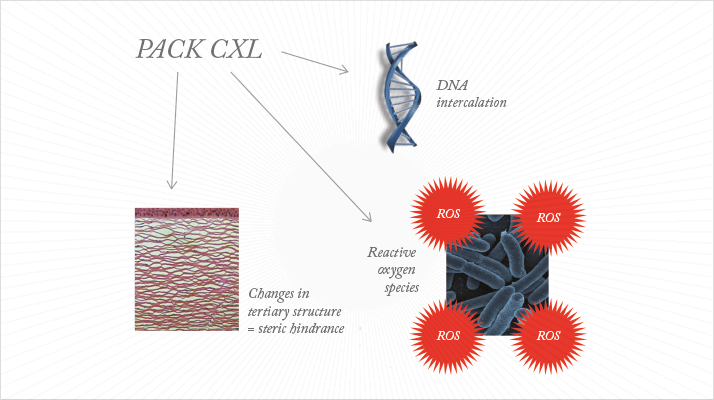
For years, transfusion medicine has exploited the antimicrobial properties of riboflavin (vitamin B2) in the presence of ultraviolet (UV)-A light (1,2). Riboflavin modifies nucleic acids on exposure to UV-A light, and when this is applied to blood, in vitro, it renders pathogens unable to replicate, in a non-toxic and non-mutagenic manner. The chromophore, UV-A-photoactivated riboflavin, is also used in ophthalmology clinics to help slow or halt the progression of keratoconus and other corneal ectasias, as it strengthens the cornea by causing oxidative corneal cross-linking (CXL). Now CXL is beginning to be used as a treatment for infectious keratitis, in a process called photoactivated chromophore for infectious keratitis-corneal cross-linking (PACK-CXL).PACK-CXL packs a triple punch (Figure 1). First is the original intended outcome of CXL: the modification of the tertiary structure of collagen. Not only does the treatment strengthen collagen, it also makes it resistant to enzymatic digestion, which means that the cornea is more resistant to the collagenases that are upregulated during bacterial invasion. Second, the process creates reactive oxygen species (ROS), which damage cell walls and are therefore cytotoxic to the pathogens. Third, the photoactivated riboflavin irreversibly intercalates with the pathogen's DNA, hindering its ability to reproduce (3).
PACK-CXL’s clinical proof of concept was demonstrated in 2008 (4). Five patients with infectious keratitis that was associated with corneal melting and who were unresponsive to topical antibiotics were treated with PACK-CXL. The progression of corneal melting was halted in all five cases and the treatment of last resort, emergency keratoplasty was avoided. Between 2008 and 2013, PACK-CXL technology was developed in vitro (6,7), in veterinary ophthalmology (8–10) and used successfully in several case series, cohort studies (11–13) and, recently, in a larger prospective study (14).
Developing country needs
Our main focus is to address unmet needs in infectious keratitis in the developing world. The challenges are patients who have:- Limited or no access to ophthalmological treatment,
- Insufficient access to medication for financial reasons and,
- Exposure to organisms that increasingly are resistant to antibiotics – although the last challenge is clearly a global one.
PACK-CXL has the potential to become a game changer on a global level, but a number of practical issues currently limit its adoption. It requires major infrastructure: bulky and expensive equipment, operated by specialized corneal surgeons in a sterile operating theater. It can be time-consuming too, requiring 10–30 minutes of treatment per session. To conquer the world, PACK-CXL needs to be delivered in a fast and inexpensive manner, without comprising patient safety. A spin-off company from the University of Geneva called EMAGine SA (www.emagine-eye.com) was created in 2013 to achieve this. The EMAGine team had four core objectives:
- The development of a small device that can be operated by the comprehensive ophthalmologist using the most basic of infrastructure in ophthalmology: the slit lamp.
- Safety mechanisms that would ensure that inexperienced users would not harm (or undertreat) the cornea by accidentally exceeding or failing to reach the required level of UV-irradiation.
- A treatment time of 5 minutes or less.
- Greater efficacy than the conventional UV light source.
- Versatility: the ability to treat keratoconus, postoperative ectasia, and pellucid marginal degeneration (PMD), if needed.
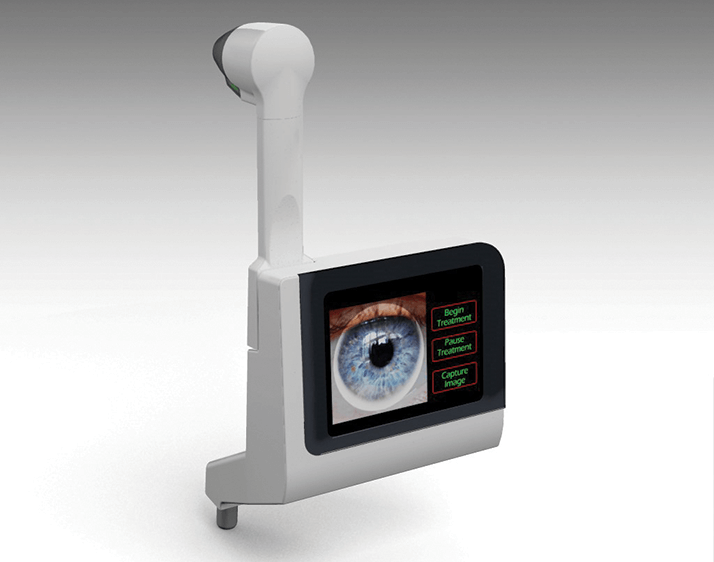
EMAGine has developed such a device: the C-Eye. It is a miniaturized irradiation system that utilizes a proprietary chromophore that can kill more than 99 percent of pathogens (bacteria and fungi) on the ocular surface in only 120 seconds. It mounts on a slit lamp in the place of the Goldmann tonometer, and can be used anywhere a slit lamp is used – why would you need a sterile operating theater to enter it with a septic case, when you can eliminate over 99% of all pathogens in two minutes? There will be two different sterile, single-use consumable tips in order to provide specific irradiation profiles, enabling the ophthalmologist to choose between a CXL tip with an irradiation profile optimized for keratoconus, PMD, and post-LASIK ectasia, or a PACK-CXL tip that provides illumination optimized for the treatment of infectious keratitis. The device has an in-built safety mechanism: real-time measurement of fluorescence of the chromophore. Whenever the fluorescence is too low (as there is not enough chromophore present) the illumination simply cuts out; the illumination also stops when the patient moves 0.5 cm away from the device. It’s easy to use. It has a touch screen, and it guides the operator throughout the procedure, and finally, it also serves as an HD video slit lamp.
These are exciting times! EMAGine believe that the C-Eye device will soon receive a CE mark, enabling its European launch this year. In summary, PACK-CXL is a promising and powerful new method that carries with it significant potential for the treatment of infectious keratitis. We hope that C-Eye will open up this treatment for every ophthalmologist across the world, in developing and developed countries alike.
Farhad Hafezi is Professor and Chairman of Ophthalmology at the University of Geneva, Switzerland, and Chief Medical and Scientific Advisor at EMAGine SA/AG, Zug, Switzerland.
References
- V. Kumar, O. Lockerbie, S.D. Keil, et al., “Riboflavin and UV-light based pathogen reduction, extent and consequence of DNA damage at the molecular level”, Photochem. Photobiol., 80, 15–21 (2004). J.P. AuBuchon, L. Herschel, J. Roger, et al., “Efficacy of apheresis platelets treated with riboflavin and ultraviolet light for pathogen reduction”, Transfusion, 45, 1335–1341 (2005). B. Tayapad, A.Q. Viguilla, J.M. Reyes, “Collagen cross-linking and corneal infections”, Curr. Opin. Ophthalmol., 24, 288–290 (2013). doi: 10.1097/ICU.0b013e32836229c5. H.P. Iseli, M.A. Thiel, F. Hafezi, et al., “Ultraviolet A/Riboflavin Corneal Cross-linking for Infectious Keratitis Associated With Corneal Melts”, Cornea, 27, 590–594. doi: 10.1097/ICO.0b013e318169d698 (2008). A. Schrier, G. Greebel, H. Attia, et al., “In vitro antimicrobial efficacy of riboflavin and ultraviolet light on Staphylococcus aureus, methicillin-resistant Staphylococcus aureus, and Pseudomonas aeruginosa” J. Refract. Surg., 25, S799–802 (2009). doi: 10.3928/1081597X-20090813-07. S.A. Martins, J.C. Combs, G. Noguera, “Antimicrobial efficacy of riboflavin/UVA combination (365 nm) in vitro for bacterial and fungal isolates: a potential new treatment for infectious keratitis.” Invest. Ophthalmol. Vis. Sci., 49, 3402–3408 (2008). doi: 10.1167/iovs.07-1592. A. Hellander-Edman, K. Makdoumi, J. Mortensen, B. Ekesten., “Corneal cross-linking in 9 horses with ulcerative keratitis”, BMC Vet. Res., 9, 128 (2013). doi: 10.1186/1746-6148-9-128. S.A. Pot, N.S. Gallhöfer, F.L. Matheis, et al., “Corneal collagen cross-linking as treatment for infectious and noninfectious corneal melting in cats and dogs, results of a prospective, nonrandomized, controlled trial”, Vet. Ophthalmol., Epub ahead of print (2013). doi: 10.1111/vop.12090. S.A. Pot, N.S. Gallhöfer, L. Walser-Reinhardt, et al., “Treatment of bullous keratopathy with corneal collagen cross-linking in two dogs”, Vet. Ophthalmol., Epub ahead of print (2013). doi: 10.1111/vop.12137. B.M. Spiess, S.A. Pot, M. Florin, F. Hafezi, “Corneal collagen cross-linking (CXL) for the treatment of melting keratitis in cats and dogs, a pilot study”, Vet. Ophthalmol., 17, 1–11 (2013). doi: 10.1111/vop.12027. K. Makdoumi, J. Mortensen, S. Crafoord, “Infectious keratitis treated with corneal crosslinking”, Cornea, 29, 1353–1358 (2010). doi: 10.1097/ICO.0b013e3181d2de91. K. Makdoumi, J. Mortensen, O. Sorkhabi, et al., “UVA-riboflavin photochemical therapy of bacterial keratitis, a pilot study”, Graefes Arch. Clin. Exp. Ophthalmol., 250, 95–102 (2012). doi: 10.1007/s00417-011-1754-1. Z. Li, V. Jhanji, X. Tao, et al., “Riboflavin/ultravoilet light-mediated crosslinking for fungal keratitis”, Br. J. Ophthalmol., 97, 669 –671 (2013). D.G. Said, M.S. Elalfy, Z. Gatzioufas, et al., “Collagen Cross-Linking with Photoactivated Riboflavin (PACK-CXL) for the Treatment of Advanced Infectious Keratitis with Corneal Melting”, Ophthalmology, Epub ahead of print (2014). doi: 10.1016/j.ophtha.2014.01.011.
