For 166 years, Bausch + Lomb has been at the forefront of technological advancement in ophthalmology – offering a wide range of eye health products across surgery, contact lenses, and lens care and pharmaceuticals. Bausch + Lomb’s Surgical International Division comprises a complete portfolio of surgical products across cataract, retinal and laser categories, with the platform’s latest arrivals reflecting the company’s ability to consistently create high-quality solutions that meet patient needs.
In cataract, the enVista® IOL platform has benefited from two recent innovations. Clinically proven since 2010, it now features an enhanced material and is available in a pre-loaded system. The aberration-free aspheric optic design also comes with StableFlex™ technology for controlled unfolding with the new SimplifEYE™ preloaded delivery system, which received the CE Mark in September 2019. The enVista IOL, with its Trusight™ optic, was the first glistening-free, singlepiece hydrophobic acrylic IOL on the market (1, 2). Available in the full diopter range, the preloaded enVista IOL is implanted through a 2.2 mm incision.
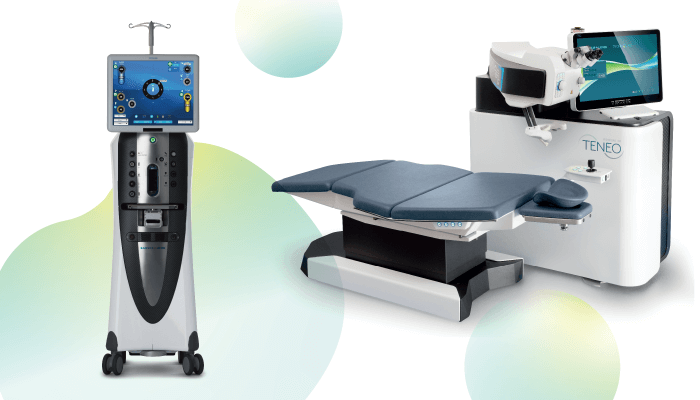
The Stellaris Elite™ vision enhancement system is amongst the latest stars to join Bausch + Lomb’s portfolio. Building on the heritage of the Stellaris® platform, the Stellaris Elite™ is a combined system that has been developed to act as the surgeon’s partner in the operating theater. “The Stellaris Elite™ is a prime example of Bausch + Lomb’s commitment to developing advanced technologies which deliver performance and efficiency to a wide range of ophthalmic procedures,” says Luc Bonnefoy, VP Surgical International at Bausch + Lomb.
Anterior features include Adaptive Fluidics™, which allows Stellaris Elite™ to automatically regulate infusion to minimize
IOP fluctuations. Moreover, active reflux enhances safety through various modes of operation. This future-proof system also features continuous irrigation activation on the wireless dual-linear foot pedal.
Looking more closely at Bausch + Lomb’s retinal innovations, the combined Stellaris Elite™ system has options for a wide range of procedural needs, including two innovative cutting technologies that take performance to another level. First, the Bi-Blade® vitrectomy cutter – with its unique dual-edge design – offers a precise and efficient cutting experience by doubling the cut rate to 15,000 cpm, designed to allow faster procedures, while potentially decreasing the risk of vitreoretinal traction (3). It also allows cutting in both forward and backwards positions, while providing consistent aspiration at all times. To complement Bi-Blade efficiency, the new cap-over configuration FreeFlow™ infusion line has been designed to facilitate a stable environment even at higher flow rates and cutting speeds. Second, the Vitesse™ hypersonic vitrectomy system represents a new approach to vitreous removal. Using patented technology, the Vitesse boasts a closed, single-needle with a small, continuously open port design. Fully open, 100 percent of the time, the Vitesse yields a virtual cut rate of approximately 1.7 million cpm.
And Bausch + Lomb’s portfolio doesn’t stop there; innovation continues into the laser category – with the TECHNOLAS® 28 Sponsored Feature Sponsored Feature 29 TENEO™ 317 Model 2 excimer being the perfect example. The company’s fastest excimer laser yet, it is able to treat at 1 s/D*, with industry-leading eyetracker technology operating at 1,740 Hz – more than three times the speed of the laser’s repetition rate. The latest software upgrade provides surgeons with an optimized workflow for the transepithelial surface ablation procedure. The transEpi PRK allows a one-step procedure to be performed without the need for alcohol by combining PTK for epithelium removal and PRK for the correction of a refractive error, for an increased ease of use and reduced treatment time. The one-step procedure also provides an optional finishing PTK – known as the “third layer” in the user interface, allowing surgeons to add treatment of up to 15 microns maximum in-depth automatically.
With new corneal procedures expected on VICTUS® Femtosecond Laser Platform in the near future, Bausch + Lomb Surgical International continues to transform the ophthalmic landscape through innovation.
For more information visit www.bauschsurgical.eu.
* Based upon calculations using an Optical Zone of 6 mm and a standard myopic treatment (PROSCAN ECO mode).
Technolas Perfect Vision GmbH is the legal manufacturer of the TECHNOLAS TENEO 317 Model 2 and the VICTUS Femtosecond Laser platform.
BLSUR_INT_INNOVADV_1219
Indications and approvals may vary by country. enVista, StableFlex, SimplifEYE, Trusight, Stellaris Elite, Adaptive Fluidics, FreeFlow, Vitesse, Technolas, Teneo and Victus are trademarks of Bausch & Lomb Incorporated or its affiliates. Bi-Blade is a trademark of Medical Instrument Development Laboratories, Inc. used by agreement. Any other product/brand names are trademarks of the respective owners.
References
- Bausch + Lomb, Internal data, enVista Direction for Use.
- P Heiner et al., “Safety and effectiveness of a single-piece hydrophobic acrylic intraocular lens (enVista®) – results of a European and Asian-Pacific study”, Clin Ophthalmol, 8, 629 (2014). PMID: 24729678.
- A Teixeira et al., “Vitreoretinal traction created by conventional cutters during vitrectomy”, Ophthalmology, 117, 1387 (2010). PMID: 20176400.
