- Trabeculectomy lowers IOP but carries significant peri- and post-operative risk
- Several techniques and devices have been developed to replace trabeculectomy
- Less invasive developments include trabecular meshwork stents and adjustable flow rate drainage devices
- Identification of the best candidate patients for each technique or device will become clearer as clinical experience grows
Surgical intervention is required under a number of circumstances in patients with glaucoma. These include unacceptable intraocular pressure (IOP) levels despite maximal topical glaucoma medication; deterioration in visual function or ocular structures, typically the optic disc; and adverse reactions (either ocular or systemic) to glaucoma medications that cannot be resolved by changing the drug or formulation.
In such cases, surgical trabeculectomy has historically been the intervention of choice – and for many patients it still is. The procedure is simple in concept – a fistula is created to connect the anterior chamber and the subconjunctival space to improve drainage – but it requires considerable skill to perform, and it also forms a bleb under the conjunctiva.
Trabeculectomy is often highly effective at reducing IOP, but the procedure is also associated with a number of complications (see “Trabeculectomy’s Troubles”), most of them related to blebbing. The shortcomings prompted the development of a range of instruments and devices that aim to either perform a better trabeculectomy, or manage glaucoma by different means.
Trabeculectomy’s Troubles
- Risks during surgery include subconjunctival hemorrhages or conjunctival buttonholes or tears.
- Early post-operative issues include filtering complications related to blebbing, such as early bleb leaks or failures, and hypotony or hypertony due to under or over-filtration.
- Late postoperative complications include many bleb-related events, such as late bleb failures or leaks, cystic bleb, blebitis, bleb dysesthesia or bleb-related endophthalmitis, plus hypotony, cataract development or progression.
Beyond trabeculectomy
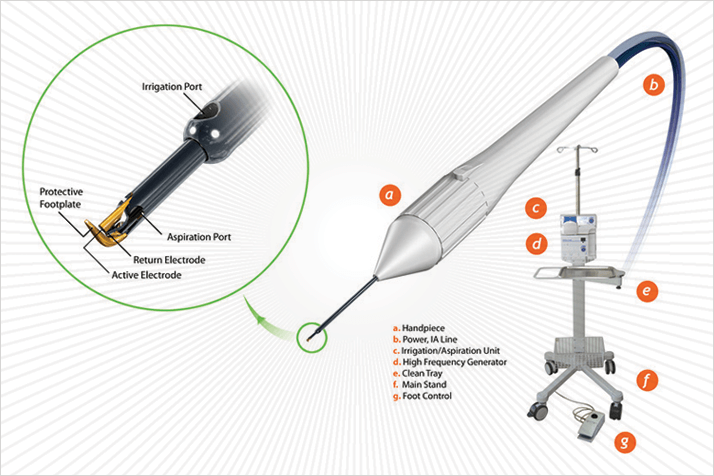
The Trabectome surgical technique is an alternative take on trabeculectomy developed by George Baerveldt at the University of California, Irvine, USA. It is an ab interno technique that uses high-frequency electrocautery to ablate a 60°–120° strip of the trabecular meshwork and the inner wall of Schlemm’s canal. The single-use Trabectome handpiece, connected to a console, performs three functions: electrocauterization; fluid irrigation, to stabilize the anterior chamber during the procedure; and aspiration, to remove the ablated tissue. The procedure impacts the area of greatest resistance to fluid outflow, removes debris and does not create a bleb. The cost of the Trabectome technique is not trivial given that, for a disposable, single-use device, it is a complex tool. It is, however, effective: data from one retrospective analysis of over 700 Trabectome procedures showed that the intervention was associated with a 40 percent decrease in IOP at 24 months after surgery (1). When combined with cataract surgery and IOL implantation, the Trabectome technique had successfully controlled IOP at one year after surgery in 87 percent of patients with exfoliation glaucoma and 91 percent of patients with primary open-angle glaucoma (2).Devices to foster fluid flow
As with trabeculectomy, the concept behind implanted drainage devices is straightforward. In this case, an apparatus that causes the aqueous humor to drain at a faster rate from the anterior chamber to the surface of the conjunctiva is placed into the trabecular meshwork, thus alleviating the increased IOP. The device may or may not have a valve. These device can be pure shunts – unvalved, pure drainage devices, or contain valves, enabling the surgeon to pre-set the drainage rate that’s required. The market for valve-containing glaucoma implant devices includes three principle products: Abbott Medical Optics’ Baerveldt device (Figure 2), New World Medica’s Ahmed valve, and Molteno Ophthalmic’s eponymous Molteno implant. Non-valved devices include Alcon’s EX-PRESS shunt (Figure 3) which, after implantation at the corneal limbus following insertion under a scleral flap, allows a limited amount of aqueous humor to drain into the intrascleral space.

Each of these devices has successfully reduced IOP in many thousands of patients with glaucoma. However, it requires skill and judgment to place drainage devices safely and correctly in order to avoid adverse events and to obtain the best possible outcomes. In particular, bleb issues – such as bleb encapsulation and conjunctival tissue thinning, and bleb infection – are not uncommon (3). New solutions are coming to the market that avoid bleb formation, such as Glaukos’ iStent (Figure 4), the iStent inject, and Ivantis Inc.’s Hydrus Microstent. These tiny devices are intended for the treatment of primary open-angle glaucoma. They are inserted into Schlemm’s canal, which they open up to let the aqueous humor drain more freely; this bypasses the blocked trabecular meshwork and all without forming a bleb. There are also bleb-less stents that utilize a different outflow pathway, namely from the anterior chamber to the suprachoroidal space. These products, such as Glaukos’ iStent Supra and Transcend Medical’s CyPass, generate potentially very large reductions in IOP.

Judging drainage perfectly is a considerable challenge, in part because intra-operative IOP can be different to pre-operative levels, depending upon the type of glaucoma the patient has, the type and location of anesthesia used, and even the procedure required to place the implant or shunt. Even the most experienced surgeon cannot predict exactly what IOP reduction might be achieved; the risk of postoperative hypertony or hypotony is ever-present. In some cases, inadequate post-operative IOP reduction can be corrected by the addition (or removal) of more shunts or stents. This, however, requires additional surgery. A drainage device could be altered for flow resistance properties after implantation without having to resort to revision surgery would be a valuable addition to the armamentarium. Such a device is now available. Called the eyeWatch (Figure 5a), it has been produced by Nikos Stergiopulos along with a team of researchers at the École Polytechnique Fédérale de Lausanne (EPFL) in Switzerland (4) for a new company, Rheon Medical. EyeWatch, which received the CE mark earlier this year, contains a drainage tube that includes a flow resistance controller. The tube can be compressed by an eccentrically shaped magnetic disk that is placed on ball bearings to allow rotation. Compression of the drainage tube, and therefore its fluidic resistance, is determined by the angle of rotation (Figure 5b). The magnetic disk can be rotated by an external control unit, providing the means of non-invasive adjustment. The device is being offered as a standalone drainage device or for use in combination with Baerveldt or Molteno implants.

The pace at which glaucoma drainage techniques and technologies are evolving is rapid and their real-world impact will become clearer as more patients receive them. Analysis of the outcomes will then allow surgeons to determine which patients should receive what device. It is likely that different approaches will serve certain types of patient better than others, depending on the type of glaucoma and IOP reduction required, but a future can be foreseen in which all but a few patients avoid trabeculectomy.
References
- D. Minckler et al., “Trabectome (trabeculectomy internal approach): additional experience and extended follow-up.”, Trans. Am. Ophthalmol. Soc., 106, 149–160 (2008). J.L. Ting et al., “Ab interno trabeculectomy: outcomes in exfoliation versus primary open angle glaucoma”, J. Cataract Refract. Surg., 38, 315–23 (2012). K.S. Schwartz, R.K. Lee, S.J. Gedde, “Glaucoma drainage implants: a critical comparison of types”, Curr. Opin. Ophthalmol., 6, 181–189 (2006). A. Villamarin et al., “A New Adjustable Glaucoma Drainage Device.”, IOVS, pii:iovs.13-12626v1 (2014). doi: 10.1167/iovs.13–12626. Epub ahead of print. Vision 2020: The Right to Sight. http://www.who.int/blindness/Vision2020_report.pdf (2007). D.A. Quillen, “Common causes of vision loss in elderly patients”, Am. Fam. Phys., 60, 99–108 (1999).
