- Current methods of glaucoma evaluation may fail patients in the early stage of the disease
- Application of OCT technology to measure multiple parameters improves the sensitivity and specificity of glaucoma diagnosis and monitoring
- In particular, measurement of the Bruch’s membrane opening-minimum rim width (BMO-MRW) can provide a clear indication of glaucomatous tissue loss, especially when it corresponds with findings from other measurements
- Use of multiple modalities to assess multiple retinal substructures may therefore assist physicians in assessing challenging patients and borderline diagnoses.
Traditionally, glaucoma diagnosis has relied on subjective assessments of optic nerve head (ONH) damage and/or measurements of visual field loss by automated perimetry. Though automated perimetry may use more objective scoring systems and algorithms, these can be unreliable – especially in the earlier, asymptomatic stages of disease, where nerve fiber loss may not translate into a detectable impairment in visual function. The “hit and miss” nature of glaucoma diagnosis results in many patients remaining unrecognized and, therefore, being deprived of vision-preserving treatments. Similarly, where diagnostic techniques are insufficiently sensitive to detect markers of disease progression, patients may not have their treatment appropriately modulated in response to advancing disease. Imaging tools and techniques able to reliably detect early glaucoma or identify patients for treatment intensification would be of great benefit to patients and public health.
Diagnosing – the problem
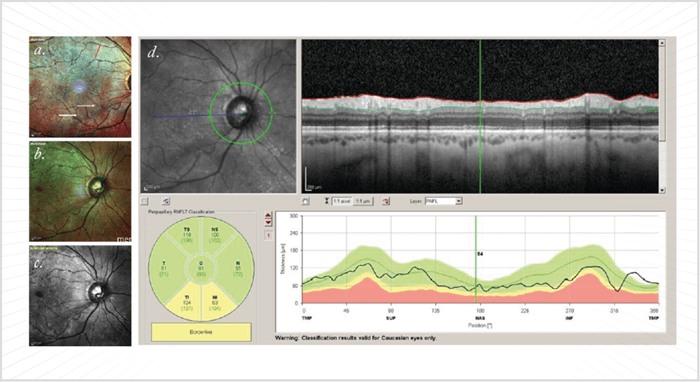
Though it is simple to spot glaucoma in patients who present with classic signals of raised IOP, optic nerve cupping and typical visual field changes, difficulties arise when the combination of these signals does not concur; for example, when IOP and visual field measurements are normal, but the optic nerve cupping is suspicious. In these cases, ophthalmologists typically employ advanced imaging technology to clarify the situation. The recognition that there was an unmet need for better glaucoma diagnosis (particularly in these challenging cases), resulted in a race to develop better imaging technologies – confocal scanning laser ophthalmoscopy, scanning laser polarimetry, and OCT. OCT is particularly useful, as it provides cross-sectional images of the ONH and macular inner retinal layers. Imaging of retinal ganglion cells (RGC) and their axons as they approach the ONH allows you to assess many parameters such as nerve fiber layer thickness and macular thickness that are highly relevant to glaucoma evaluation – the macula has the highest RGC concentration of all retinal areas, and so cell loss is more readily detectable in this region. OCT-based measurements can also assist differential diagnosis. Other pathologies that can masquerade as glaucoma include brain tumors, strokes, and even aneurysms pressing on the optic tract – all of which are serious conditions needing urgent attention. In neurological abnormalities, the pattern of visual loss respects the vertical midline in both eyes – a clear pointer that additional investigations are required to rule out a neurological cause. Less serious conditions that may interfere with diagnosis include retinal scars in the periphery, vascular occlusions in the retina, a tilted optic nerve, or even epiretinal membranes (Figure 1). However, there are still difficulties with OCT. Diagnoses can be missed because changes indicative of genuine glaucomatous damage to the optic nerve may be ascribed to “green disease” if the loss is focal or an artifact such as an epiretinal membrane hides the loss of tissue thickness. “Red disease” on the other hand is artifact-related OCT abnormalities in the absence of real glaucomatous damage. These misdiagnoses arise because normal anatomic variation in optic nerve structure is not yet fully reflected in the normative databases available to OCT devices. For example, highly myopic eyes often have atypical optic nerves – unusually sized, or associated with atypical features such as epiretinal membranes, tilted discs, peripapillary atrophy, or traction artifacts (Figure 2). Incorrect glaucoma diagnoses in these in patients leads to unnecessary medication or surgery, and the consequences can be lifelong; even if misdiagnosed patients present for a second opinion, it is difficult to suggest ceasing treatment because longitudinal records of IOP, OCT or visual field readings often are lacking, making it impossible to confirm or rule out disease progression. However, I firmly believe that over- or under-diagnosis can be avoided by employing multiple modalities to measure different anatomical features.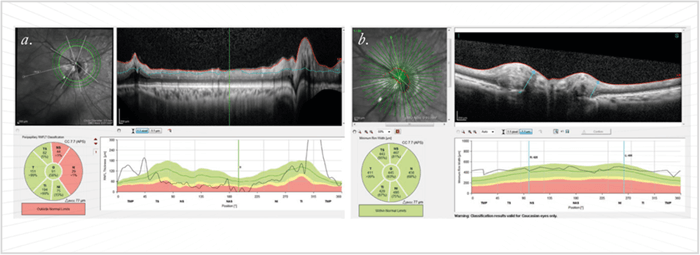
The spectra of modalities and measurements
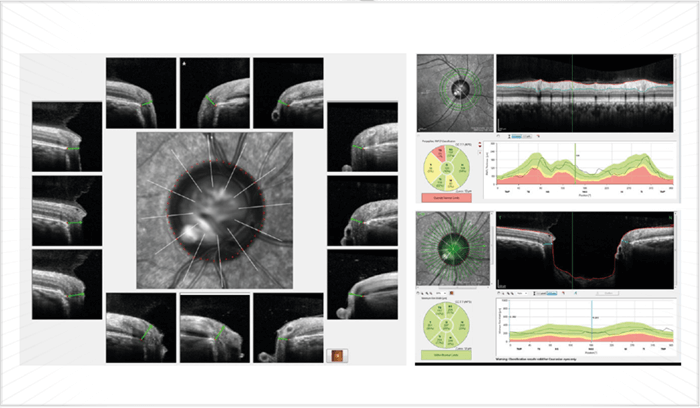
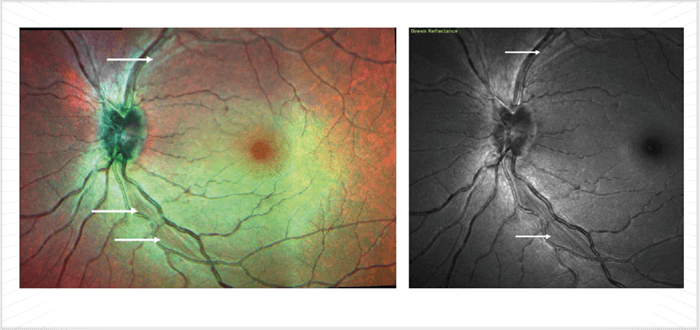
As OCT device resolution has improved over the years, it has allowed segmentation of individual layers and recognition of anatomical features, such as the Bruch membrane opening (BMO). Similarly, successive software modifications have extended OCT capabilities from measurement of retinal nerve fiber layer (RNFL) thickness to ONH evaluation – and now to segmentation of the macular inner retinal layers (for example, with Topcon’s DRI-OCT Atlantis or Heidelberg Engineering’s Spectralis). Such measurements allow automatic calculation of multiple modalities at once, such as RNFL thickness, the Bruch’s membrane opening-minimum rim width (BMO-MRW – the thickness of the nerve tissue lining the optic canal), and macular thickness. Once artifacts are excluded, advanced OCT systems are very useful for monitoring stability in early and moderate glaucoma. The BMO-MRW may be particularly useful for clarifying borderline abnormal results by helping confirm the presence or absence of RNFL abnormalities in the presence of surface pathologies, such as peripapillary epiretinal membranes (which distort RNFL thickness measurements, see Figure 3), or in patients with tilted myopic optic nerves and severe peripapillary atrophy. BMO-MRW is reported to be a better criterion for detecting early glaucoma than either circumpapillary RNFL thickness or Bruch membrane opening-horizontal rim width (BMO-HRW): at 95 percent specificity, the sensitivity was 81 percent, 70 percent and 51 percent for BMO-MRW, RNFL thickness and BMO-HRW respectively (1). In short, including BMO-MRW assessments should be beneficial for glaucoma diagnosis. It should be noted that in late glaucoma, tissue thickness is so minimal that RNFL and BMO-MRW measurements are of little value; however, macular thickness can still be used to monitor the disease state in these patients. For analysis of BMO-MRW, normative databases only exist for the Heidelberg Spectralis OCT system. However, the Spectralis has no normative databases for the assessment of macular ganglion cells,– unlike Zeiss’ Cirrus and the Optovue’s RT-Vue. As ganglion cells – analysis can avoid artifacts arising where total retinal thickness is affected by non-glaucomatous disease, instruments with this capability have advantages over the Spectralis. However, though segmentation capabilities of various devices are improving, such software is still subject to significant errors. I also measure asymmetry between the eyes and hemispheres, which is offered on the Spectralis. The concept here is that the findings should correspond; for example, tissue loss indicated by RNFL and BMO-MRW measurements should match with a decrease in macular thickness and when compared with the other eye, the changes should be asymmetric as is typical of mild to moderate glaucoma. Such correspondence gives much more confidence in a glaucoma diagnosis; if the three do not correspond, it may well not be glaucoma (Figure 4). The Spectralis machine is also unique in that it has the capability of multicolor, i.e., it can interrogate the retina with infrared, blue and green wavelengths, and then combine these three outputs to provide multicolor images of the fundus; the blue and green wavelengths depict the RNFL in exquisite detail. This modality combined with OCT can be very useful for early detection of RNFL loss (which is to say, before the peripapillary thickness has reduced to abnormal values; Figure 5). In such cases, the presence of arcuate RNFL loss can confirm very early glaucoma – even in the absence of classic signals. Moreover, the multicolor modality is useful for improving medication compliance, as the images can make tissue loss evident to patients, motivating them to adhere to their treatment regimen. Without these pointers, the lack of subjective symptoms – particularly in early or even moderate glaucoma – results in patients failing to appreciate the need for medication.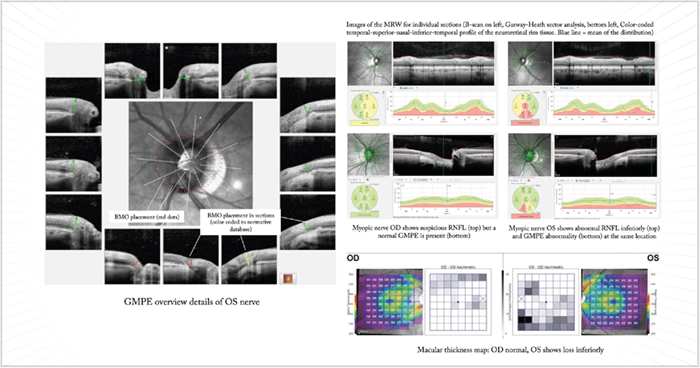
A measure of success
In summary, measurements of the macular inner retinal layer are useful for diagnosing early glaucoma and monitoring subsequent disease progression so as to guide treatment – but are not so useful for patients who have advanced glaucoma and/or coexisting retinal disease. Modern OCT is very useful for analyzing BMO parameters, and it’s possible that BMO-MRW will become the dominant disc parameter for OCT-mediated glaucoma diagnosis; however, we should remember that there can be significant age-related reduction in BMO-MRW measures in healthy subjects (2), so it will be important to adjust for age when using this measure. In my practice, I find SD-OCT very useful; multimodal imaging, eye-tracking functionality, and multicolor scanning capabilities can all assist in glaucoma diagnosis and management. I have found multicolor imaging particularly useful in borderline diagnostic cases (Figure 4). Modern systems clearly have a key place in today’s glaucoma practice. Sanjay Asrani is a glaucoma specialist at Duke Eye Center at Cary, Cary, NC, USA.References
- BC Chauhan et al., Ophthalmology 120, 535–543 (2013). PMID: 23265804. BC Chauhan et al., Ophthalmology, 122, 1786–1794 (2015). PMID: 26198806.
