
I’ve been performing canaloplasty for nearly two decades. I started off with the ab externo surgical technique, but always wished for a minimally invasive way of rejuvenating the natural outflow pathway. And that’s why I teamed up with Nova Eye Medical to develop the ab interno surgical technique back in 2014. Since then, I’ve performed tens of thousands of ab interno canaloplasty procedures, and now teach the procedure to residents, fellows, and colleagues at the Dean McGee Eye Institute at the University of Oklahoma. The ab interno canaloplasty method is now my go-to MIGS – and the new iTrack™ Advance is a major development in this field.
The iTrack™ Advance offers the same technical and clinical advantages as the original iTrack microcatheter, including the illuminated tip and the unique viscoelastic injector. The former gives visual verification of the microcatheter tip location at all times – critical to confirm the device has remained within Schlemm’s canal rather than entering the suprachoroidal space or a collector channel. The viscoelastic injector permits administration of precise volumes of viscoelastic, up to 100 microliters, depending on surgeon preference; this ability to titrate the delivery of viscoelastic enables us to take a truly nuanced treatment approach for each and every patient. Crucially, the comprehensive approach of canaloplasty addresses the entire outflow system, including the collector channels. This is an important distinction of canaloplasty, when you consider that other MIGS procedures address only a couple of clock hours of the conventional outflow pathway; remember that we still have no diagnostic that defines the site of canal constriction, so localized treatment is not an option.
Easier for new users
iTrack™ Advance has equivalent safety and effectiveness to the original iTrack; indeed, the main differences relate to how easily it can be adopted into a surgeon’s treatment paradigm. The original procedure required needle-based goniotomy, needle removal, forceps use, and catheter insertion. Next, the surgeon was required to feed the microcatheter through the main incision, often traversing the pupil to reach all the way across to the angle. Some residents and fellows found this approach challenging, and the elimination of these steps with iTrack™ Advance is very welcome. Dispensing with the need for needle and forceps makes the technique much easier for new users. Notably, for standalone procedures, the new iTrack™ Advance only requires a 1.0 incision, which is sufficient for anterior chamber access, microcatheter insertion, progression through 360 degrees, and egress.
A more subtle usability feature comes in the form of iTrack™ Advance’s rotatable nozzle, which allows both left- and right-handers to conveniently and precise position the cannula, as required.
Finally, the new actuator mechanism on the handpiece uses a 2:1 gear ratio to prevent unwanted movement – critical when undertaking surgery next to the cornea.
Those new to iTrack™ Advance can see some of its advantages for – one of many benefits showcased in the video is the phenomenal efficiency of the procedure.
 Mahmoud A. Khaimi discussing the next generation canaloplasty device, iTrack™ Advance, during the ESCRS 2022, Milano, Italy.
Mahmoud A. Khaimi discussing the next generation canaloplasty device, iTrack™ Advance, during the ESCRS 2022, Milano, Italy.
Together, these advantages make the procedure uniquely efficient: once you engage the actuator, the microcatheter just shoots out – it’s the Ferrari of MIGS! – but in a safe and precisely controlled manner.
The iTrack™ Advance Combines Proven Benefits of iTrack with New Features and a Simpler Technique
Original iTrack:
200-micron illuminated canaloplasty microcatheter: continuous visual confirmation of tip location
handheld injector: actuator-driven advance of microcatheter, precise titration of administered viscoelastic
The new iTrack™ Advance:
spatulated cannula tip: facilitates insertion into TM, access to Schlemm’s canal
adjustable nozzle: rotatable to suit left- or right-handed use
actuator mechanism: advance/withdraw the microcatheter without the need for forceps
simpler technique
Trusted, with a serious makeover
The original iTrack is backed up by over 100,000 procedures performed over 10 years of global use. And the same three-year data – from over a thousand eyes, treated by multiple surgeons – that supports the original iTrack also apply to the iTrack™ Advance. The new device retains the same proprietary features, including the illuminated tip that lends iTrack a clear safety advantage. In short, iTrack™ Advance is evidently just as safe and effective as the original – both when used as a standalone procedure and when combined with cataract surgery – but we’ve given it a serious makeover. It’s now more ergonomic, more efficient, and easier to use.
iTrack™ Advance represents a new route into the advantages of canaloplasty; namely, the comprehensiveness nature of approach, which acts on distal collector channels, the TM, and Schlemm’s canal.
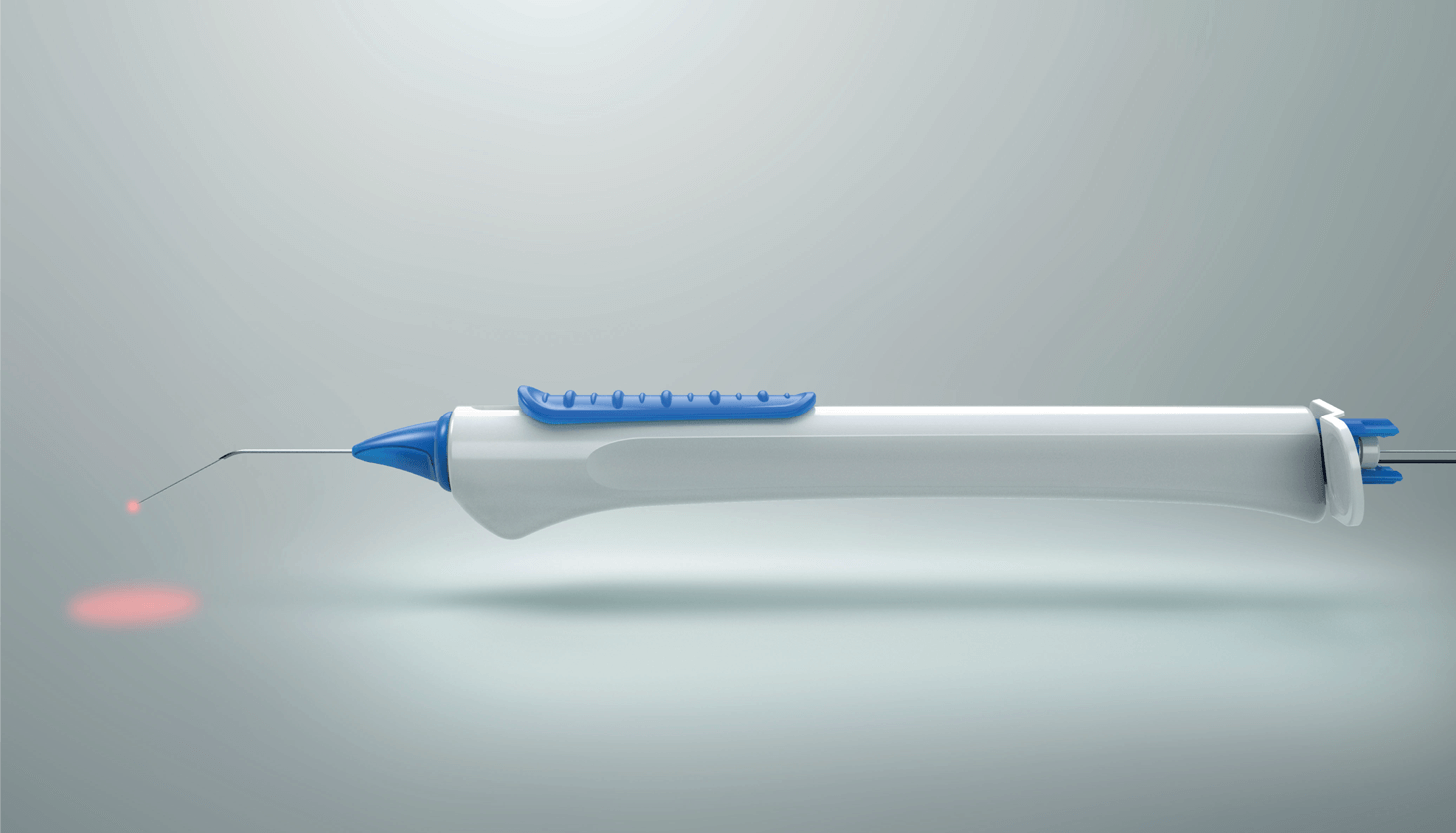 Track™ Advance canaloplasty device (Nova Eye Medical)
Track™ Advance canaloplasty device (Nova Eye Medical)
Watch a panel of experts discuss the iTrack Advance and canaloplasty
Learn more about iTrack™ Advance
iTrack™ Advance has been cleared for the indication of fluid infusion and aspiration during surgery, and for catheterization and viscodilation of Schlemm’s canal for the reduction of intraocular pressure (IOP) in adult patients with open-angle glaucoma. iTrack™ Advance has a CE Mark (Conformité Européenne) for the treatment of open-angle glaucoma. iTrack™ Advance is not available for use or sale in the USA
