A Consultant Ophthalmologist since 2006, James Ball has for the last six years owned Custom Vision Clinic – a private practice specialising in refractive and premium cataract surgery. One key driver for Mr. Ball in establishing his clinic was to explore the potential of Implantable Collamer Lens surgery.
Mr. Ball wanted the flexibility to set the price of ICL surgery at a level that would be attractive to patients, while still being profitable. He began offering ICLs as a refractive surgery option alongside corneal laser refractive surgery – ensuring that the cost gap between the two was small enough to give patients a real choice in selecting the procedure most clinically beneficial for them. “The conversion rate for patients to whom I’ve recommended ICL surgery is close to 100 percent,” Mr. Ball tells The Ophthalmologist.
“Having access to ICL surgery ensures that we’re not tempted – as we might have been – to push the limits of corneal laser refractive surgery,” he explains. “Whether it’s PRK, SMILE, or LASIK, the risks of ectasia (1, 2, 3), dry eye syndrome (4), and night-vision disturbance, (5,6,7) all increase after corneal laser refractive procedures with those larger myopic corrections. ICLs offer an alternative that can be a great option for patients with thin corneas (8), they do not induce dry eye syndrome (9), and they can provide patients with exceptional vision day and night (10,11). In terms of safety, STAAR Surgical’s EVO family of lenses “really stand alone” in the arena of phakic IOL technology, says Mr. Ball. “In the last 20 years, I’ve seen multiple phakic IOL designs come and go because of issues with safety.” The biocompatibility of the Collamer material, however, has really stood the test of time; “none of my ICL patients have experienced any long-term complications following implantation.”
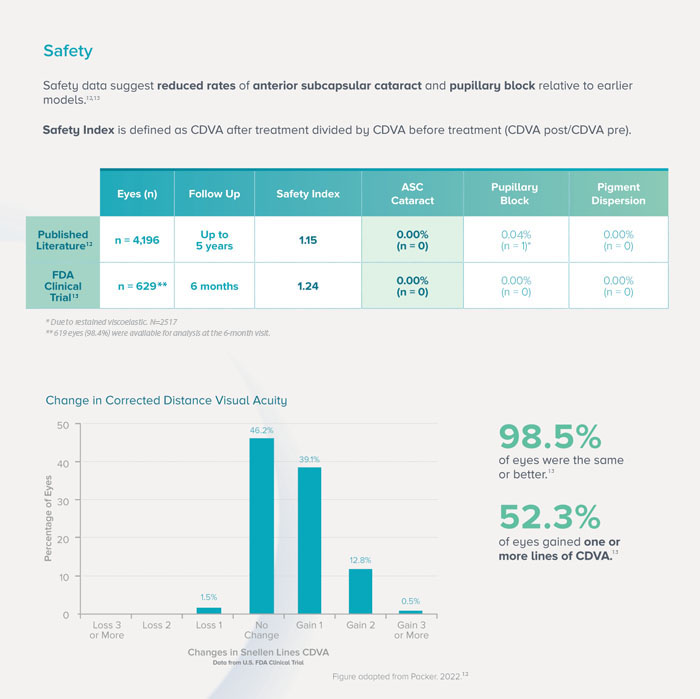 Figure 1. Credit: Image sourced from STAAR Surgical EU EVO HCP Brochure, 2022 STAAR ® Surgical Company - MKT-0482-EU-ENG REV. 1 (12, 13).
Figure 1. Credit: Image sourced from STAAR Surgical EU EVO HCP Brochure, 2022 STAAR ® Surgical Company - MKT-0482-EU-ENG REV. 1 (12, 13).
In determining who is suitable for the EVO ICL, Mr. Ball follows the guidance provided by STAAR Surgical regarding the magnitude of refractive error. He discusses ICL implantation as an option for all refractive surgery candidates, explaining where it fits into their particular recommendation. “For example, if I have a minus four myope with a thinner than average cornea and/or risk factors for significant dry eye syndrome, I will explain the pros and cons of both corneal laser refractive surgery and ICL implantation, but will likely recommend ICL for the excellent visual quality and minimal postoperative dry eye symptoms with no risk of ectasia (14).”
 Figure 2. Credit: Image sourced from STAAR Surgical EU EVO HCP Brochure, 2022 STAAR ® Surgical Company - MKT-0482-EU-ENG REV. 1 (12, 13, 15, 16).
Figure 2. Credit: Image sourced from STAAR Surgical EU EVO HCP Brochure, 2022 STAAR ® Surgical Company - MKT-0482-EU-ENG REV. 1 (12, 13, 15, 16).
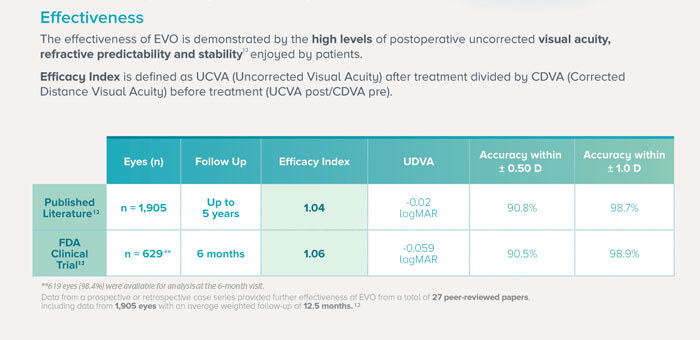 Figure 3. Credit: Image sourced from STAAR Surgical EU EVO HCP Brochure, 2022 STAAR ® Surgical Company - MKT-0482-EU-ENG REV. 1 (12, 13, 15, 16).
Figure 3. Credit: Image sourced from STAAR Surgical EU EVO HCP Brochure, 2022 STAAR ® Surgical Company - MKT-0482-EU-ENG REV. 1 (12, 13, 15, 16).
Figure 2. Credit: Image sourced from STAAR Surgical EU EVO HCP Brochure, 2022 STAAR ® Surgical Company - MKT-0482-EU-ENG REV. 1 (12, 13, 15, 16).
Figure 3. Credit: Image sourced from STAAR Surgical EU EVO HCP Brochure, 2022 STAAR ® Surgical Company - MKT-0482-EU-ENG REV. 1 (12, 13, 15, 16).
Not only is the ICL a useful tool for the correction of primary refractive errors, it has also been shown to be effective as a piggyback lens for correcting the refractive error in pseudophakic patients, notes Mr. Ball (17). Regarding the presbyopic population he states “for whatever reason, these patients may not be suitable for corneal laser refractive surgery. But with EVO Viva ICL, which provides increased depth of focus, there is now an option for the correction of presbyopia in our older myopic patients. This is really exciting because it means removing fewer clear crystalline lenses from a myopic patient population that is at an increased risk of retinal detachment following lens replacement (18).”
For Mr. Ball, the EVO ICL strengthens his service offering by providing patients not just the latest diagnostic technology and surgical techniques, but also the safest possible options. “For patients, it’s all about the result,” he says. “And in gaining spectacle independence with great quality of vision and minimal side effects, the vast majority of my ICL patients are delighted with the outcome of their treatment.”
Important Safety Information for the EVO ICL Family
The EVO/EVO+ ICL is indicated for phakic patients 21-60 years of age, and pseudophakic patients 21 years and older, to correct/reduce myopia up to -20.0 D with up to 6.0 D of astigmatism. The EVO Viva ICL is indicated for phakic patients 21-60 years of age, and pseudophakic patients 21 years and older, to correct/reduce myopia up to -20.0 D with or without presbyopia. Careful preoperative evaluation and sound clinical judgment should be used by the surgeon to decide the risk/ benefit ratio before implanting a lens in a patient with any of the conditions described in the DFU. Prior to surgery, physicians should inform prospective patients of possible risks and benefits associated with the EVO/EVO+ ICL or EVO Viva ICL. Reference the EVO/EVO+ ICL and EVO Viva ICL DFUs available at https://edfu.staar.com/edfu/ for a complete listing of indications, contraindications, warnings and precautions.
References
- MA Wolle et al., “Complications of refractive Surgery: Ectasia after Refractive Surgery,” Int Ophthalmol Clin, 56, 129, (2016). PMID: 26938343.
- MI Asif et al., “Complications of small incision lenticule extraction,” Indian J Ophthalmol, 68, 2711, (2020). PMID: 33229647.
- G Pushpanjali and D.T. Azar, “Risk Profiles of Ectasia after Keratorefractive Surgery,” Curr Opin Ophthalmol, 28, 337, (2017). PMID: 28594648.
- B Sharma et al., “Impact of corneal refractive surgery on the precorneal tear film,” Indian J Ophthalmol, 68, 2804, (2020). PMID: 33229655.
- DPS O’Brart et al., “Disturbances in Night Vision After Excimer Laser Photorefractive Keratectomy,” Eye, 8, 46, (1994) PMID: 8013719.
- C Villa et al., “Night vision disturbances after successful LASIK surgery,” Br J Ophthalmol, 91, 1031, (2007). PMID: 17314153.
- M Li et al., “Effects of Wavefront Aberrations on Night Vision Problems and Mesopic Contrast Threshold After SMILE,” J Refract Surg, 37, 446, (2021). PMID: 34236902.
- GD Parkhurst et al., “Phakic intraocular lens implantation in United States military warfighters: a retrospective analysis of early clinical outcomes of the Visian ICL,” J Refract Surg, 27, 473, (2011). PMID: 21243974.
- S Ganesh, et al., “Matched population comparison of visual outcomes and patient satisfaction between 3 modalities for the correction of low to moderate myopic astigmatism,” Clin Ophthalmol, 11, 1253, (2017). PMID: 2874036.
- E Martínez-Plaza et al., “Effect of the EVO+ Visian Phakic Implantable Collamer Lens on Visual Performance and Quality of Vision and Life,” Am J Ophthalmol, 226, 117, (2021). PMID: 33577790.
- GD Parkhurst, “A prospective comparison of phakic collamer lenses and wavefront-optimized laser-assisted in situ keratomileusis for correction of myopia,” Clin Ophthalmol, 10, 1209, (2016). PMID: 27418804.
- M Packer, “The Implantable Collamer Lens with a central port: review of the literature,” Clin Ophthalmol, 12, 2427 (2018). PMID: 30568421.
- M Packer. “Evaluation of the EVO/EVO+ Sphere and Toric Visian ICL: Six moth results from the United States Food and Drug Administration clinical trial,” Clin Ophthalmol, 16, 1541 (2022). PMID: 35645557.
- R Wei, et al., “Comparison of objective and subjective visual quality early after implantable collamer lens V4c (ICL V4c) and small incision lenticule extraction (SMILE) for high myopia correction,” Acta Ophthalmol, 98, 943, (2020). PMID: 32419383.
- G Schild et al., “Uveal and capsular biocompatibility of a single-piece, sharp edged hydrophilic acrylic intraocular lens with collagen (Collamer®): 1-year results,” J Cataract Refract Surg, 30, 1254 (2004). PMID: 15177600.
- DC Brown, SL Ziémba, “Collamer® intraocular lens: clinical results from the US FDA core study,” J Cataract Refract Surg, 27, 833 (2001). PMID: 11408127.
- JF Alfonso et al., “Implantable Collamer Lens for Management of Pseudophakic Ametropia in Eyes With a Spectrum of Previous Corneal Surgery,” J Refract Surg, 34, 654, (2018). PMID: 30296326.
- V Daien et al., “Incidence, risk factors and impact of age on retinal detachment following cataract surgery in France: a national population study,” Journal of the American Academy of Ophthalmology, 122, 2179, (2015). PMID: 26278859.
