Béatrice Cochener-Lamard, Head Ophthalmology Department, Brest University Hospital, France
Marc Labetoulle, Chairman and Professor of Ophthalmology, Head Department of Ophthalmology, Paris-Sud University, France
Rita Mencucci, Professor, School of Ophthalmology, University of Florence, Italy
Moderator: Rudy Nuijts, Professor of Ophthalmology, Vice-Chairman and Director Cornea Clinic and the Center for Refractive Surgery, University Eye Clinic, Maastricht University Medical Center, Netherlands
On Friday, October 2, the European Society of Cataract and Refractive Surgeons (2020 Congress) hosted a virtual symposium intended for ophthalmologists and surgeons: “Improving the Patient Experience in Cataract Surgery.” The meeting was chaired by Rudy Nuijts, who opened proceedings by noting the importance of optimizing patient care while reducing healthcare costs (1).
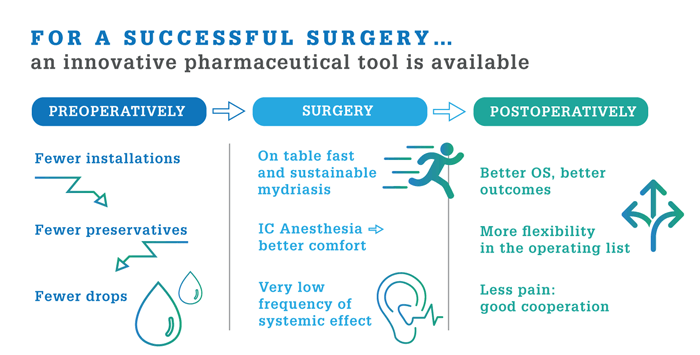
Prof. Labetoulle reminds us that fast-track cataract removal (topical anesthesia, outpatient setting) reduces risk, stress, and discomfort for the patient. Moreover, it minimizes patient time in the clinic – especially important during the COVID-19 pandemic. Data suggest speed and comfort may be further improved by adoption of Mydrane® solution for injection (Laboratoires Théa) *, an innovative, intracamerally-injected mydriasis product (2).
"Mydrane® is the first choice for about 20 percent of French cataract surgeries." Marc Labetoulle
Prof. Labetoulle summarizes a retrospective comparison of Mydrane® and conventional mydriasis eye drops (3): “Mydrane® reduced surgery time by 20 percent, room occupancy time by 12 percent, patient turnaround time by 30 percent, and increased daily throughput from 10 procedures to 12 procedures.” Total time-saving was six minutes; at a cost of €11/minute for a surgery room, Mydrane® therefore saves ~€60 per procedure.
"Mydrane® improves management of cataract surgery – better patient care for less cost." Marc Labetoulle
Theory supports the above findings: Mydrane® adoption models indicate that a center performing 3,000 cataract surgeries per year would reduce waiting times by 1,500 hours per year, save 500 hours per year of nurse time (by removing the need for repeated eyedrop instillation), and save 40 hours of surgeon time per year – which equates to an extra four sessions of surgery annually (4). The exact figures and cost-savings, Prof. Labetoulle notes, will vary from country to country.
"In France, private clinics routinely use Mydrane®, which suggests it is financially advantageous." Marc Labetoulle
In summary, Prof. Labetoulle notes that Mydrane® can decrease patient turnaround time, reduce nurse and surgeon time, and save costs. Furthermore, it does not compromise safety and efficacy: “Visual outcomes are unaffected, and ocular surface abnormalities are reduced at one and four months.”
Prof. Cochener-Lamard states patient satisfaction after cataract surgery is affected by waiting time (5), pain management, and visual outcomes – all of which benefit from Mydrane®. Firstly, Mydrane® reduces preoperative clinic time by avoiding repeated eyedrop instillation. Secondly, Mydrane® intra-ocular anesthesia both reduces pain and also provides efficient mydriasis, thereby making surgery safer and more comfortable, improving patient cooperation. Thirdly, by avoiding preservative-containing mydriatic eye drops, Mydrane® reduces the risk of ocular surface disease exacerbation and poor visual outcomes**.
“Mydrane® improves patient experience in three important areas – waiting time, pain management, and visual outcomes.” Béatrice Cochener-Lamard
Prof. Mencucci offers five pointers to an improved patient experience:
- Pre-operative communication: if the patient has OSD, tell them! Remember that about half of all cataract patients have ocular surface abnormalities – and most of those are asymptomatic (6).
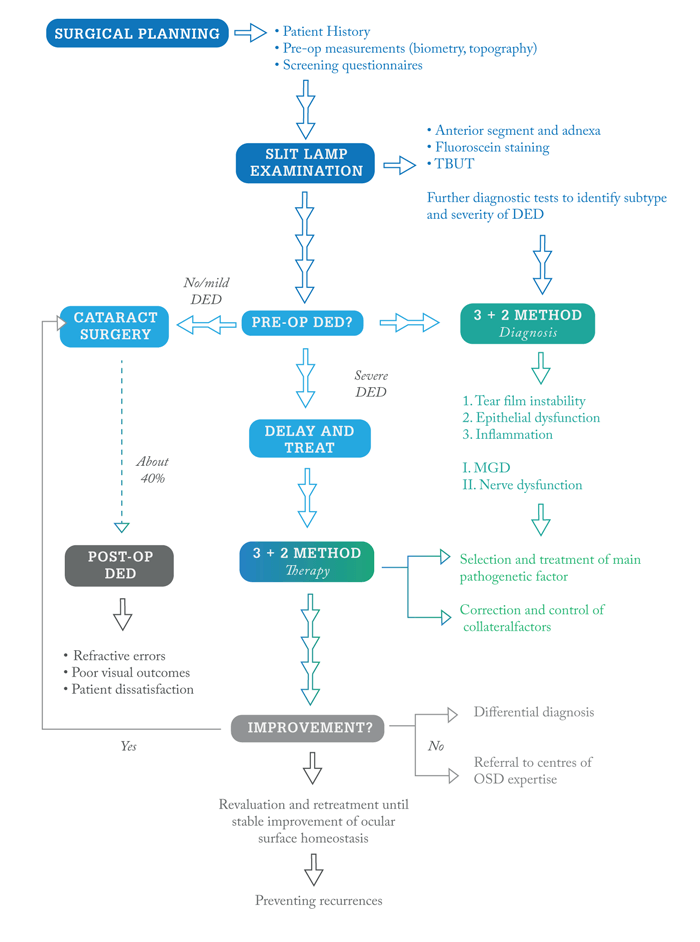
- Diagnosis and planning: use accepted algorithms (7) to guide management and stabilization of OSD.
- Optimize the ocular surface before surgery: Treat EBMD and Salzmann degeneration before surgery to avoid irregular astigmatism; consider preoperative superficial keratectomy (PDK); use artificial tears; treat inflammation and infection; avoid preservative-containing eye drops.
- Preserve the ocular surface during surgery: consider smaller incisions, more rapid surgery, and lower illumination intensity. Use polymeric corneal coating instead of surface irrigation (8); limit the use of anesthetics and mydriatic eye drops. Of note, data show that Mydrane®, but not the standard protocol, is associated with complete recovery of tear break-up time within 15 days (9).
- Support the ocular surface after surgery: choose preservative-free artificial tears. Avoid NSAIDs and antibiotics in eyes with visual disturbances, rheumatological symptoms, severe OSD, or premium IOLs.
“Replacing topical medications with Mydrane® helps preserve the ocular surface during cataract surgery.” Rita Mencucci
Given productivity advantages and the avoidance of increased pain commonly associated with a second operation, Prof. Nuijts proposes that bilateral cataract removal will soon become more common. Prof. Nuijts concisely summarizes the discussion by saying that, in some environments, intracameral anesthesia and dilatation may enhance patient experience, preserve the ocular surface, and reduce costs.
*MYDRANE®, Solution for injection. Tropicamide 0.2 mg/mL, phenylephrine hydrochloride 3.1 mg/mL, lidocaine hydrochloride 10mg/mL. Indicated for cataract surgery to obtain mydriasis and intraocular anaesthesia during the surgical procedure in adults only. Mydrane® should only be used in patients who have already demonstrated, at pre operative assessment, a satisfactory pupil dilation with a topical mydriatic therapy. For more information, see SmPC.
** Very few adverse effects have been registered since the launch of the product. For detailed information, see SmPC.
References
- ME Porter, EO Teisberg, “Redefining Healthcare,” Harvard Business Review Press: 2006.
- JL Guell et al., “Towards a better cataract surgery efficiency with intracameral injection of Mydrane® Oral communication.” Presented at the ESCRS Winter Meeting; 2018; Belgrade, Serbia.
- W Salah et al., “Assessment of the duration of surgery and patient turnover after the incorporation of a standardised intracameral combination of mydriatics and anaesthetics for cataract surgery,” J Cat Refract Surg, 46, 1487 (2020). PMID: 32675648.
- K Davey et al., “Budget impact model of Mydrane®, a new intracameral injectable used for intra-operative mydriasis, from a UK hospital perspective,” BMC Ophthalmol, 18, 104 (2018). PMID: 29673317.
- M McMullen, P Netland, “Wait time as a driver of overall patient satisfaction in an ophthalmology clinic,” Clin Ophthalmol, 7, 1655 (2013). PMID: 23986630.
- PK Gupta et al., “Prevalence of ocular surface dysfunction in patients presenting for cataract surgery evaluation,” J Cat Refract Surg, 44, 1090 (2018). PMID: 30078540.
- CE Starr et al., “An algorithm for the preoperative diagnosis and treatment of ocular surface disorders,” J Cat Refract Surg, 45, 669 (2019). PMID: 31030780.
- R Mencucci et al., “Triphasic polymeric corneal coating gel versus a balanced salt solution irrigation during cataract surgery,” J Cat Refract Surg, 45, 1148 (2019). PMID: 31371007.
- R Mencucci et al., “Protecting the ocular surface at the time of cataract surgery: intracameral mydriatic and anaesthetic combination versus a standard topical Protocol,” Ophthalmol Ther. 2020 Oct 14. PMID: 33052582.
