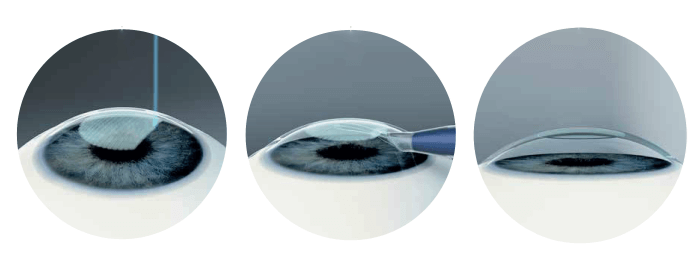
The XENIA Implant is unique in the advanced keratoconus treatment space. Boasting increased stiffness, the XENIA material regularizes corneal topography and reduces higher order aberrations. Unlike other implants that are constructed from plastic or metal, the XENIA uses natural corneal collagen – the “real thing” – and so it is very well tolerated by the patient corneas. Thanks to an additive approach, the procedure is fully reversible (unlike subtractive procedures), which makes it an attractive alternative to corneal transplantation. The procedure itself is also simple – a stromal pocket is created in the cornea by Femtosecond laser and a customized implant is inserted into the pocket, where it stabilizes the patient’s cornea and helps improve their vision. The secret behind XENIA’s effectiveness lies in its design; each device is custom-made, manufactured upon prescription for each individual patient, and XENIA’s Technical Documentation is fully compliant with the latest EU Medical Device Regulation MDR 2017/745.
In the surgeon’s hands: Ahmed Elmassry, Cornea and Refractive Surgeon, Professor and Head of Ophthalmology Department, Faculty of Medicine at Alexandria University, Egypt
Treating patients with advanced keratoconus has always been a challenge. Patients typically need keratoplasty or corneal surgery; long, and difficult procedures with a
steep learning curve and a high risk of postoperative complications. There are also problems with availability of grafts, sutures, and general anesthesia, as well as quality of vision, and cost issues. I began using the XENIA implant 11 months ago, and 90 percent of my patients have been satisfied with the result. Most have reached BCVA of 0.5 and have stable vision after six months.
Implantation technique
The procedure itself is simple. I create a femtosecond laser-assisted pocket with a diameter of 7 mm of the optical zone, and an incision of 5.5 mm using the Zeiss VisuMax machine (I use the SMILE parameters for -0.25 D, but without removing the lenticule). Then, I insert the folded implant in the pocket as I would for the DMEK graft. It is a 10-minute procedure, similar to refractive laser in terms of preparation, sterilization, and the use of local anesthetic eye drops. It is sutureless, which facilitates corneal recovery, and causes no immunological reaction. It’s also low risk, low cost, and requires fewer drugs postoperatively. Best of all, it is pain-free, which gives patients a better experience.
A better option
I consider the implant to be an effective alternative to DALK corneal transplant – it is certainly more standardized. DALK comes with significant risks: the fluctuating cost and availability of donor corneas, a steep learning curve, the risk of graft rejection, and the need for postoperative drugs for nine months to one year after surgery.
XENIA, on the other hand, is totally reversible and practically pain-free. The implant has a very fast recovery time, similar to that of a refractive procedure, where DALK takes around two weeks. This is in part because the XENIA implant uses natural collagen rather than plastic or metal, which means better tolerability and no rejection or reaction – you can even crosslink the cornea after the procedure.
Tailor-made geometry
Another benefit of the XENIA implant is that you can tailor its geometry to each individual cornea, which can improve visual outcomes. XENIA is also made from material stiffer than the regular cornea and DALK corneas, which are prone to develop ectasia. The stiff implant regulates irregular corneas, resulting in better topography after surgery, fewer high order aberrations (HOA), and better visual acuity.
The implant works best for patients with advanced keratoconus, who have no scars or previous hydrops. I would not recommend it for early keratoconus patients who would be better treated with corneal cross-linking (CXL), moderate cases that need CXL and ICRS, or very advanced cases with broken endothelium or corneal scars. In a study, we crosslinked six out of seven XENIA implants after three months of surgery. XENIA’s additive thickness enables CXL that may not be possible in thin corneas.
In the surgeon’s hands: Balasubramaniam Ilango, Consultant Ophthalmic Surgeon specializing in Corneal Transplant and Laser Refractive surgery at the Royal Wolverhampton Hospitals NHS Trust, UK and Medical Director at Optimax Clinics, UK
I treat all kinds of corneal disorders, including keratoconus, Fuchs’ corneal endothelial dystrophy, and herpetic corneal disease. Advanced keratoconus is a complex eye condition that cannot be treated with contact lenses or spectacles. Often the cornea is too thin for cross-linking, leaving a donor transplant the only option. Such surgery takes two hours to perform under general anesthesia and requires frequent follow-up visits. Visual recovery can be slow due to the presence of corneal sutures and induced astigmatism. Patients are often quite young when this happens and, unfortunately, have to put their life on hold for several years.
I have been using the XENIA implant since November 2019, and my experience has been very encouraging. It is truly a groundbreaking invention. Following implantation of the lenticule, I have found the average corneal thickness increased from 401 μm to 513 μm. Average optical K readings were not statistically altered (51.4 D versus 51.5 D), while anterior corneal astigmatism decreased from 7.4 D to 2.0 D. Unaided vision improved from 1.74 LogMar to 1.54 LogMar. The implant provides a fully reversible additive, thickening of the cornea.
Reducing the risk
In my opinion, the XENIA implant is more than an effective alternative option to DALK surgery. DALK carries risk of graft rejection, suture related infection and astigmatism, as well as the risks associated with general anesthesia. The XENIA implant is comparatively risk-free.Moreover, it is reversible and can be completed in under 30 minutes (DALK surgery usually takes two to three hours). The majority of my XENIA patients returned to work on day two, whereas my DALK patients need a minimum of two weeks’ time off work, and up to three months off from normal activities.
For example, one of my patients underwent LASIK surgery in 2009, but developed post-LASIK ectasia in his left eye with poor vision (1.30 LogMar) seven years later. He didn’t want to proceed with human donor corneal transplant surgery due to the risks. Fortunately, his right eye vision was good enough to carry on with his employment. He had XENIA implant surgery in his left eye in November 2019. His unaided vision has improved to 0.70 LogMar postoperatively and he is now enjoying binocular vision.
Avoiding corneal transplantation is certainly advantageous, particularly as people are living longer; often, donor organs have low endothelial cell counts, leading to graft failure within 10 years. And that means further corneal transplant operations with additional risks...
Material properties
One clear benefit of the XENIA implant is the material used. INTACS/Cornea ring segments made from plastic materials have a high risk of extrusion and poor patient tolerance. However, XENIA implants have the potential to integrate into the recipient cornea as they are constructed from biological collagen. XENIA implants are also stiffer than human donor organs and – as a result – yield a better corneal configuration, fewer HOA, and better visual acuity after surgery. I also like how the XENIA implant is unique to each patient; everyone’s corneal geometry/topography is different, and so a custom-made XENIA implant can correct the individual patient’s corneal deficiency, leading to the best possible visual outcome.
Corneal implants and the pandemic
Finally, the XENIA implantation surgery is an outpatient procedure – and that seems more important than ever. In a post- COVID-19 environment, reducing the number of health professionals needed for a procedure – anesthetists in particular – will help maintain better social distancing. And let’s not forget the possibility that human donor organs can carry the SARS-CoV-2 virus. Until this can be ruled out, it would be better to avoid human donor grafts. (In the UK, all elective solid organ and corneal transplant procedures have been stopped temporarily.) XENIA implants appear to be a blessing in the COVID-19 environment.
I remove epithelium from the patient’s eye prior to pocket formation. I then use the Intralase Femtosecond Laser to create a custom corneal stromal pocket of 100 μm depth and 8.7 mm diameter, with a 3.7 mm access tunnel. The pocket was designed so once opened with a pocket dissector, it would accept a 120-μm 7.2 mm diameter XENIA lenticule.
My pearls for surgeons:
• Remember to adjust the head posture during femto-docking in
patients with deep-set socket/prominent nose
• Preoperative tetracaine 1% topical anesthesia would help to debride the corneal epithelium easily
• Take care while dissecting the stromal bed as the cornea is quite thin; avoid perforating it. Ask the patient to look down while inserting the dissector
• 7+ mm diameter XENIA implant would be easier to insert, cover the visual axis
• Avoid touching the XENIA implant with cotton buds to reduce impurities
• Hold the XENIA implant with non-toothed straight forceps and insert into the pocket with a single gentle, slow motion.
Gebauer Medizintechnik GmbH: Germany.
