
Heidelberg Engineering burst onto the OCT scene back in 2007 with the SPECTRALIS® diagnostic imaging platform – a powerful combination of confocal scanning laser ophthalmoscopy (cSLO), spectral domain OCT (SD-OCT) and patented eye-tracking technology that has set the standard for multimodal retinal imaging for over a decade. Having explored the potential of swept-source OCT technology (SS-OCT) since 2011, the company entered into a patent license agreement with Massachusetts General Hospital in Boston, USA, in 2015, granting global and exclusive rights to 77 basic patents and patent applications which relate to SS-OCT technology and its application in ophthalmology. Developing both of these OCT technologies side-by-side for the last seven years, the company has carefully assessed the advantages and limitations of each technology in order to capitalize on their strengths by optimizing each for specific ocular imaging applications.
Clinical benefit first and foremost
Heidelberg Engineering has always felt a strong sense of responsibility to deliver technical innovations with strong clinical benefits, as well as to preserve the continuity of patient data in both the research and clinical settings. Since the commercial release of SS-OCT in 2012, clinical research has been comparing SD-OCT and SS-OCT technologies applied to retinal imaging. Evaluating divergent research studies for years, Heidelberg Engineering has found no confirmation that the longer wavelength employed by commercial SS-OCT devices provides clinically significant information beyond that of SD-OCT imaging. In fact, some research has shown that deep ocular structures, such as the choroidal/scleral interface, can be equally visualized by SS-OCT and by SD-OCT using enhanced-depth imaging (EDI) (1).. SS-OCT devices once offered speed advantages over that of SD-OCT. However, advancements in both OCT technologies have kept pace, with current SD-OCT devices being faster with reported speeds up to 250 KHz (2). While SS-OCT devices use a longer wavelength to better penetrate media opacities, Heidelberg Engineering has been combining the benefits of confocality and OCT automatic real-time image averaging to successfully minimize the impact of media opacities for over 10 years. The clinical advantages of the SS-OCT angiography technique also remain to be seen. At the same time, the potential to further enhance the axial resolution of the SD-OCT images, which is not currently possible for SS-OCT, could play a significant role in the future clinic.
The value of data continuity
Since continuity of data is vital to accurate assessment of disease progression, Heidelberg Engineering has and will continue to optimize SD-OCT technology for the posterior segment. The SPECTRALIS OCT2 Module was designed to keep pace with advancements in speed and depth penetration without sacrificing data continuity and image quality. Shorter wavelengths provide higher contrast for inner retinal layers. The company’s patented active eye-tracking technology continues to advance, allowing for image averaging up to 100 times within an entire volume scan. The higher contrast is further appreciated as a result of image averaging due to increased signal-to-noise ratio.SS-OCT for the anterior segment
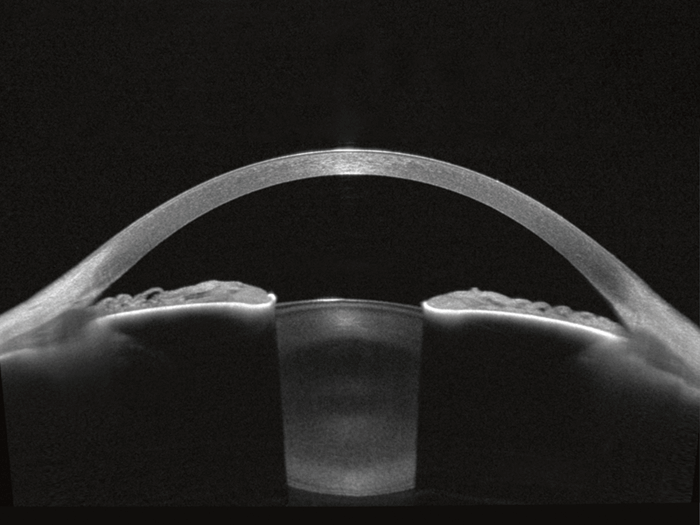
While SS-OCT technology has not revolutionized retinal imaging, the higher light output at the longer wavelength of 1,300 nm results in the best imaging conditions for the anterior segment of the eye. Heidelberg Engineering has optimized SS-OCT technology to combine all relevant anterior segment examinations in one modular, upgradeable platform. The ANTERION®* offers topography, tomography, anterior segment metrics, and axial length in one device. The stunning SS-OCT images provide visual confirmation of the reliability and accuracy of the measurements.
Conclusion
Heidelberg Engineering’s optimization of SS-OCT technology for anterior segment imaging and SD-OCT technology for retinal imaging will continue to deliver images that empower eye care professionals to make confident diagnostic decisions that will ultimately improve patient care. *Please note that ANTERION is not available for sale in the United States at this time.
References
- SM Waldstein et al., Eye, 29, 409–415 (2015). PMID; 25592119. L An et al., Biomed Opt Express, 2, 2270–2283 (2011). PMID: 22025983.
