Recently, Alcon achieved 100 million implantations of their AcrySof® platform, one of the world’s most frequently implanted IOLs (1). But there is always room for improvement, and as cataract surgery constantly advances and evolves, two key areas of innovative focus are the IOL design and biomaterial, and preloaded delivery devices. What improvements would cataract surgeons like to see in IOLs? High biocompatibility, greater optical clarity, as well as a simplified and easy to use IOL injector that delivers reproducible results. Alcon – a company with a proven track record of developing and manufacturing best-in-class IOLs – has a new offering based on this feedback: Clareon® AutonoMe™, the next generation of preloaded IOLs.
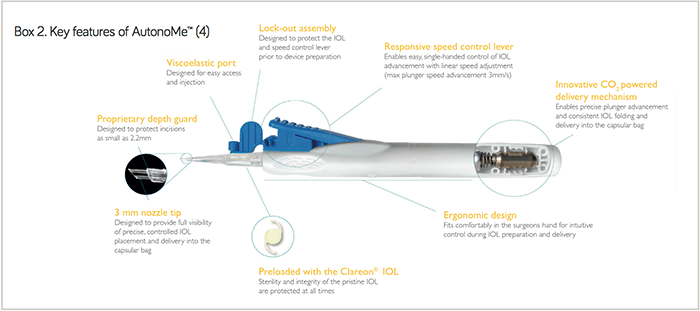
Clareon® (Box 1) is an advanced monofocal IOL with all the same features of the trusted AcrySof® platform. However, a new hydrophobic acrylic biomaterial and an enhanced manufacturing process have enabled further innovation, such as the precision edge design, which reduces edge glare (2)(3). Clareon® allows surgeons to provide unsurpassed clarity for their patients with no haze, subsurface nanoglistenings or glistenings, all with a low incidence of posterior capsule opacification (4)(5)(6)(7)(8). Accompanying Clareon® is AutonoMe™ (Box 2) – the next evolution of IOL delivery from Alcon. AutonoMe™ is the first and only automated, disposable, pre-loaded IOL delivery system in the market, and uses an innovative CO2-powered delivery mechanism that allows surgeons easy – and predictable – control over IOL delivery and speed. Not only that, it is simple to use.
Key BioOptics features
- Precision edge design to minimize potential for edge glare and positive dysphotopsias (2,3)
- Fully usable 6 mm biconvex aspheric optic (9)
- UV and blue light filtering (4)
Key BioMechanics features
- STABLEFORCE® Haptics remain planar during unfolding
- Rapid and controlled unfolding (10)
- Precision edge design that guards against PCO and minimizes Nd:YAG procedures, in addition to reducing edge glare (3,4)
New BioMaterial features
- Advanced manufacturing process
- Among the lowest level of surface haze, subsurface nanoglistenings (SSNGs), and glistenings of competitive monofocal IOLs (5–8)*
- Automated, disposable, preloaded IOL delivery system
- Innovative CO2- powered lens advancement with a responsive speed control lever (4)
- Linear control of IOL delivery speed up to 3.0 mm/s (4)
- Easy, intuitive and control
Rudy Nuijts, Professor of Ophthalmology and Director of the Cornea Clinic, Maastricht University Medical Center, Maastricht, The Netherlands “We have been implanting Clareon® lenses in patients since July 2017, and we’ve found that the Clareon® IOL handles less ‘stiffly’ during the cartridge manipulation and loading than the AcrySof® lens, and that there is a quicker unfolding of the optic and haptics in the eye. We also found less ‘stickiness’ of the haptics on the optic, as you can sometimes see with the AcrySof®. We identified a four second unfolding difference between the Clareon® and AcrySof® lenses (30 ± 13.5 s vs, 34 ± 15.7 s, respectively).” Stephen Lane, Clinical Professor of Ophthalmology at the University of Minnesota and Medical Director of Associated Eye Care in Stillwater, Minnesota, USA; and Chief Medical Officer of Alcon “AutonoMe™ allows single-handed delivery, and utilizes a very innovative, automated delivery system with a CO2-powered cartridge and responsive speed control lever that allows the surgeon to stop or start – and vary the speed of – the delivery. All surgeons like control throughout all phases of the surgery, and AutonoMe™ enables precise delivery into the capsular bag, whilst protecting incisions as small as 2.2 mm and providing full IOL visibility during the delivery. I believe it will change the way which we deliver IOLs into the eye.”
References
- Alcon. “Alcon flagship AcrySof® intraocular lens reaches 100 million implants and counting”. Available at: http://bit.ly/2hqAnYT. Accessed October 31, 2017. Improved Milled Edge Quality of Clareon® IOLs. Alcon internal technical report: TDOC-0053598, effective March 13, 2017. Schematic Model Eye and In Vitro Evaluation of Positive Dysphotopsia or Glare Types Photic Phenomena. Alcon internal technical report: TDOC-0053578, effective August 8, 2017. Clareon® AutonoMe™ Directions for Use. Microvacuole Evaluation of Clareon® and Other Marketed IOLs. Alcon internal report: TDOC-0053516, effective February 28, 2017. Clareon® and Marketed IOL Slit Lamp Surface Haze. Alcon internal technical report: TDOC-0053487, effective February 28, 2017. Clareon® and Marketed IOL Nidek Surface Scatter and Bulk Haze. Alcon internal technical report: TDOC-0053488, effective February 28, 2017. Elimination of Clareon® IOL Surface Haze Summary. Alcon internal technical report TDOC-0053072, effective 05-Jan-2017. Imaging of the Usable Optic Diameter of Clareon® SY60WF, TECNIS ZCB00, and enVista MX60 IOLs. Alcon internal technical report: TDOC-0053803, effective July 12, 2017. L Werner and N Mamalis. Evaluation of IOL positioning stability and ocular biocompatibility of a new IOL material in the rabbit cataract surgery model (University of Utah) – Interim Report.
