
Sobha Sivaprasad presents data supporting the use of corticosteroids for DME insufficiently responsive to anti-VEGF agents
Contains promotional information by Alimera Sciences. Please click here to view the prescribing information and adverse event reporting information for ILUVIEN® (fluocinolone acetonide [FAc]).
A presentation given by Sobha Sivaprasad at the Royal College of Ophthalmologists (RCOphth) annual congress 2015 entitled “Role of steroid in diabetic macular oedema” described the pivotal role of corticosteroids in the management of chronic diabetic macular edema (DME). The talk highlighted the efficacy and tolerability of corticosteroids, and the RCOphth recommendation for the use of these agents in center-involving chronic DME not sufficiently responsive to anti-vascular endothelial growth factor (anti-VEGF) agents.1
Inflammation is central to DME pathophysiology and, as Professor Sivaprasad explained, is a cause rather than a consequence of the disease. Indeed, the hyperglycemia associated with uncontrolled diabetes is directly responsible for inflammation within the eye, which leads to a breakdown of the blood–retinal barrier, Müller cell dysfunction, and microglial activation, resulting in the alteration of retinal homeostasis and photoreceptor dysfunction.1 Therefore, it is important to consider inflammatory pathways when treating DME.
Corticosteroids—both licensed such as fluocinolone acetonide (ILUVIEN®, Alimera Sciences) and dexamethasone (Ozurdex®, Allergan Inc.) , and unlicensed, such as triamcinolone (Trivaris, Allergan Inc.)—are typically used as second-line therapies in DME. Data demonstrate that the efficacy of corticosteroids in reducing central retinal thickness (CRT) is comparable to that of anti-VEGF agents (for example, Lucentis®; Novartis).2 The reduction in retinal thickness with both therapies has shown to be greater in patients with a CRT≥400 µm than in patients with a CRT≤400 µm.3
Professor Sivaprasad went on to present data from the “Macular Edema: Assessment of Implantable Dexamethasone in Diabetes (MEAD)” study of Ozurdex and the “Fluocinolone Acetonide in Diabetic Macular Edema (FAME)” study of ILUVIEN, which are indicated in the treatment of chronic DME and DME, respectively.
Macular Edema: Assessment of Implantable Dexamethasone in Diabetes (MEAD)—An Ozurdex study

The MEAD study included 1,048 patients with DME who were randomized to receive DEX implant 700 µg (licensed dose of Ozurdex), DEX implant 350 µg or sham. The progress of these patients was followed for three years. Those who required repeat treatment had to wait a minimum of six months.1 The mean number of treatments received over three years was 4.1, 4.4, and 3.3 with DEX implant 700 µg, DEX implant 350 µg, and sham, respectively (Figure 1).5
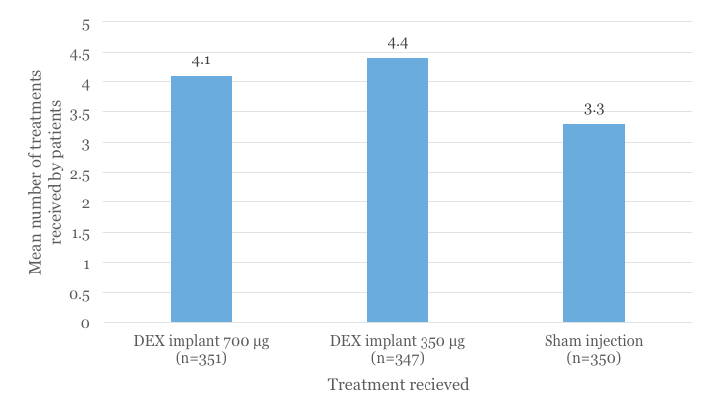
Figure 1. Mean number of treatments received by patients over a three year period in the MEAD study.5
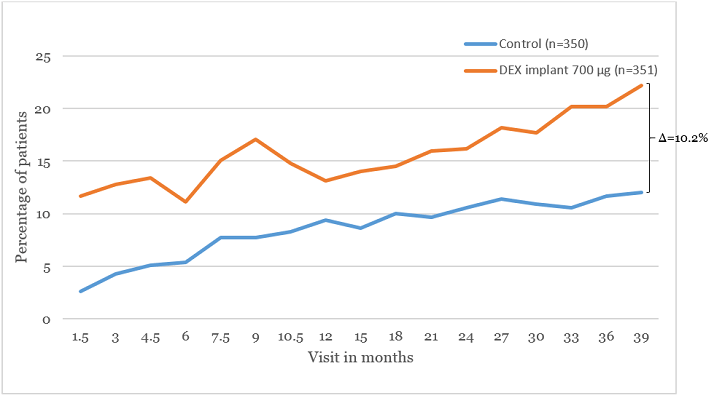
The percentage of patients with ≥15-letters best-corrected visual acuity (BCVA) improvement was significantly higher with DEX implant 700 µg (22.2 percent) compared to sham (12.0 percent) at the Year 3 visit (Figure 2).6 During the study period, the mean change in BCVA from baseline was 3.5 letters with DEX implant 700 µg, 3.6 with the DEX implant 350 µg and 2 letters with sham.5
Figure 2. Proportion of patients in the MEAD study with ≥15-letters improvement in BCVA. Adapted from the European Medicines Agency, Committee for Medicinal Products for Human Use Ozurdex Assessment report (EMA/492068/2014).6
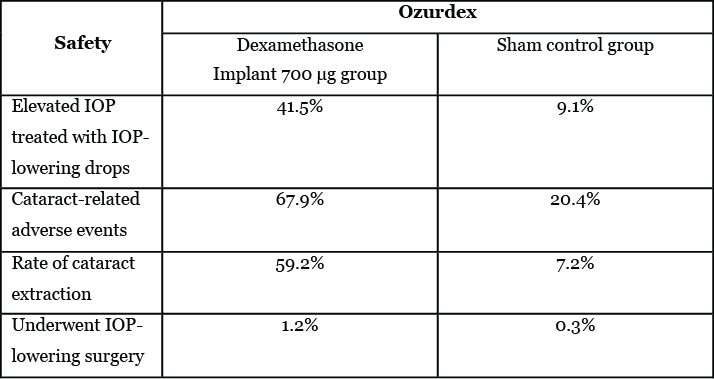
Patient treatment with DEX implant 700 µg resulted in the greatest mean average reduction in CRT from baseline (-111.6 µm), followed by DEX implant 350 µg (−107.9 µm) and sham (−41.9 µm; P<0.001). Rates of cataract-related adverse events in phakic eyes were 67.9 percent, 64.1 percent, and 20.4 percent in DEX implant 700 µg, DEX implant 350 µg, and sham groups, respectively. Regarding intraocular pressure (IOP) incidences requiring procedural intervention—such as trabeculectomy, irisdectomy and iridotomy—there were four cases of such incidences in the DEX implant 700 µg group (Table 1).5
Table 1. IOP and cataract incidence in the dexamethasone implant 700 µg group compared to the sham control group in the MEAD study.5
Fluocinolone Acetonide in Diabetic Macular Edema (FAME)—An ILUVIEN study

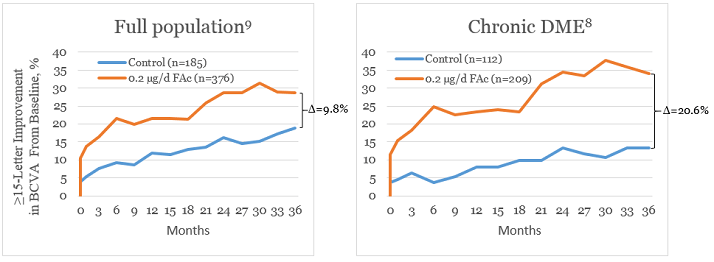
The use of ILUVIEN in DME is supported by data from the FAME study, in which 953 patients were randomly assigned to treatment with 0.2 µg FAc (ILUVIEN, licensed dose), 0.5 µg FAc or sham. Over three years of treatment, ILUVIEN led to significantly more patients achieving a ≥15-letter improvement in BCVA compared to sham (Figure 3). Importantly, ILUVIEN was more effective in patients with chronic DME, which is the approved indication in Europe. The rate of cataract extraction with ILUVIEN was 80 percent in the overall DME population (27.3 percent in the sham group) and 85.1 percent in the chronic disease patient group (36.4 percent in sham group) (Table 2).1,8,9
Table 2. IOP and cataract incidence in the 0.2 µg ILUVIEN FAc implant group compared to the sham control group in the FAME study.8,10
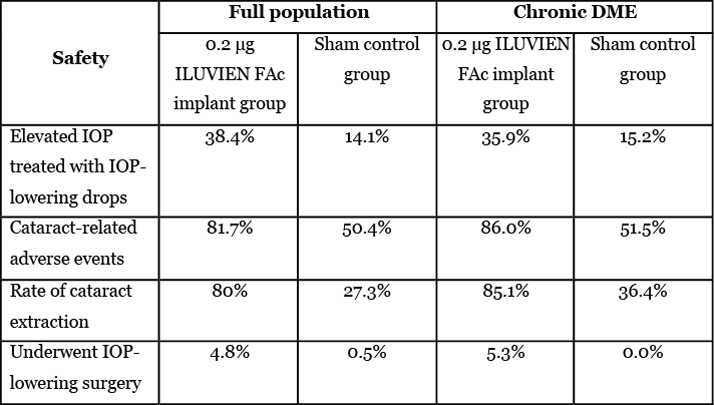
Figure 3. Proportion of patients with a ≥15-letter improvement in BCVA with ILUVIEN and sham treatment in the FAME study.8,9
Based on the available data, the RCOphth recommends that corticosteroids should be used as second-line therapy for center-involving DME, where treatment with laser or anti-VEGF agents has led to an insufficient response (Figure 4).1,11 This recommendation falls within the guidelines from the National Institute for Health and Care Excellence (NICE), which place no restriction on CRT thickness for ILUVIEN use, and state that ILUVIEN should be used in chronic DME in pseudophakic patients insufficiently responsive to available therapies.13 NICE has estimated that 26.2 percent of DME patients have a pseudophakic lens and that 60.6 percent of patients overall have chronic DME.14 NICE has recently updated its guidance for Ozurdex which was published after the RCOphth annual congress 2015; however, data from the guidance have been included in this article for completeness.15
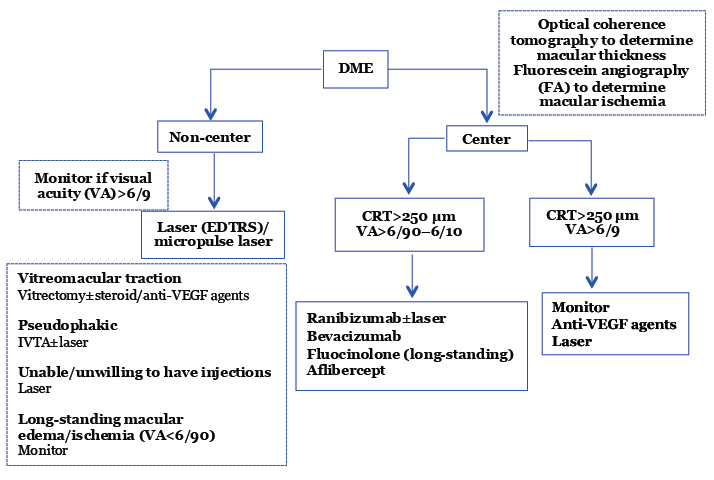
Figure 4. Recommendation for the treatment of DME according to the RCOphth.11
Professor Sivaprasad emphasized the importance of switching patients with an insufficient response from anti-VEGF agents to corticosteroids. Following 12 months of monthly anti-VEGF injections, 50 percent of patients had persistent macular thickening.16 Given NICE guidance, pseudophakic patients are obvious candidates for a switch to corticosteroids.1
The presentation by Professor Sivaprasad reminds ophthalmologists that DME is an inflammatory disease and should be considered accordingly. Corticosteroids have shown to be efficient and well-tolerated in DME treatment, with ILUVIEN showing better outcomes in chronic DME treatment. The RCOphth guidelines recommend that phakic and pseudophakic patients with center-involving DME are appropriate for therapy with corticosteroids, such as ILUVIEN, when insufficiently responsive to other treatments, in eyes where CRT is 250 µm or above.17 Professor Sivaprasad concluded by recommending the use of corticosteroids, particularly triamcinolone, in DME patients with a CRT≥400 µm.1
Sobha Sivaprasad is Professor of Retinal Clinical Studies at the Institute of Ophthalmology, University College London. She is a Consultant Ophthalmologist at Moorfields Eye Hospital, specializing in medical retina. Her higher research degree was on age-related macular degeneration (AMD). She obtained her Medical Retina Fellowship from Moorfields Eye Hospital. Dr Sivaprasad has active clinical and laboratory research interests in AMD, diabetic retinopathy and retinal vascular disorders. Her research focuses on biomarkers and modeling of retinal morphology in retinal diseases. She oversees several multicenter clinical trials in these areas and has over 100 peer-reviewed publications to her credit. She is an Editor of two eye journals and peer-reviewer of several ophthalmic journals.
Look out for DME content developed by Alimera Sciences on this website throughout 2015. We hope it supports your knowledge of DME and ILUVIEN, and if you would like to contribute material for publication, please send your materials to dmecontenthub@hayward.co.uk, we’d be very pleased to consider your contributions.
REFERENCES 1. S Sivaprasad, “Role of steroid in diabetic macular oedema”, presented at the Royal College of Ophthalmologists Annual Congress 2015, 19–21 May, Liverpool, UK, (2015). 2. W Amoaku, “A review of therapies for diabetic macular oedema and rationale for combination therapy”, Eye, 1–16 (2015). PMID: 26113500. 3. The Diabetic Retinopathy Clinical Research Network, “Randomized Trial Evaluating Ranibizumab Plus Prompt or Deferred Laser or Triamcinolone Plus Prompt Laser for Diabetic Macular Edema”, American Academy of Ophthalmology, 117(6), 1064–1077, e35, (2010). PMID: 20427088. 4. Electronic Medicines Compendium, “Ozurdex summary of product characteristics’, 92015). Available at: https://www.medicines.org.uk/emc/medicine/23422. Accessed July 2015. 5. D Boyer et al., “Three-Year, Randomized, Sham-Controlled Trial of Dexamethasone Intravitreal Implant in Patients with Diabetic Macular Edema”, Ophthalmology, 121(10), 1904–1914 (2014). PMID: 24907062. 6. European Medicines Agency, Committee for Medicinal Products for Human Use (CHMP), “Ozurdex Assessment report (EMA/492068/2014)”, (2014). Available at: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Assessment_Report_-_Variation/human/001140/WC500173294.pdf. Accessed August 2015. 7. Electronic Medicines Compendium, “ILUVIEN summary of product characteristics”, (2015). Available at: https://www.medicines.org.uk/emc/medicine/27636. Accessed July 2015. 8. P Campochiaro et al.,” Sustained Delivery Fluocinolone Acetonide Vitreous Inserts Provide Benefit for at Least 3 Years in Patients with Diabetic Macular Edema”, Ophthalmology, 119(10), 2125–2132 (2012). PMID: 22727177. 9. P Campochiaro et al.,”Long-term Benefit of Sustained-Delivery Fluocinolone Acetonide Vitreous Inserts for Diabetic Macular Edema”, Ophthalmology, 118(4), 626–635 (2011). PMID: 21459216. 10. J Cunha-Vaz, “Sustained Delivery Fluocinolone Acetonide Vitreous Implants: Long-Term Benefit in Patients with Chronic Diabetic Macular Edema”, Ophthalmology, 121(10), 1892–903 (2014). PMID: 24935282. 11. F Ghanchi, Diabetic Retinopathy Guidelines Working Group, “The Royal College of Ophthalmologists’ clinical guidelines for diabetic retinopathy: a summary”, Eye, 27, 285–287 (2013). PMID: 23306724. 12. Data on File. Alimera Sciences. 13. National Institute for Health and Care Excellence, “Fluocinolone acetonide intravitreal implant for treating chronic diabetic macular oedema after an inadequate response to prior therapy (rapid review of technology appraisal guidance 271)”, 2013. Available at: https://www.nice.org.uk/guidance/ta301. Accessed June 2015. 14. National Institute for Health and Care Excellence Costing template, Dexamethasone intravitreal implant for treating diabetic macular oedema (TA349). (2015) Available at: https://www.nice.org.uk/guidance/ta349/resources. Accessed August 2015 15. National Institute for Health and Care Excellence, “Dexamethasone intravitreal implant for treating diabetic macular oedema (technology appraisal guidance TA349)”, 2015. Available at: https://www.nice.org.uk/guidance/ta349. Accessed August 2015. 16. A Das et al., “Diabetic Macular Edema: Pathophysiology and Novel Therapeutic Targets”, Ophthalmology, 122, 7, 1375–1394 (2015). PMID: 25935789. 17. The Royal College of Ophthalmologists, “Diabetic retinopathy guidelines”, (2012, updated 2013). Available at: https://www.rcophth.ac.uk/standards-publications-research/clinical-guidelines. Accessed July 2015.
UK-ILV-MMM-0269 Date of preparation: July 2015
Founded in 2003, Alimera Sciences researches and develops innovative vision-improving treatments for chronic retinal diseases, such as diabetic macular edema (DME), dry age-related macula degeneration (AMD), and retinal vein occlusion. In 2015, Alimera Sciences partnered with The Ophthalmologist to facilitate the publication of independently created educational content surrounding DME, a serious retinal complication associated with diabetes, which is increasing in incidence with the increasing prevalence of diabetes worldwide. Published content will include articles ranging from basic science and disease processes to overviews of clinical data, different surgical procedures, comparisons of treatment options, and practical advice for managing diabetic patients. With a commitment to honesty, integrity, responsibility, candor, and trust, Alimera Sciences intend to provide educationally focused content to healthcare professionals across a wide range of topics in DME in order to both increase disease awareness and understanding, and to help improve patient outcomes. UK-ILV-MMM-0355 Date of preparation: August 2015 enquiries@alimerasciences.com

