
Educational content provided by Alimera Sciences.
Diabetic macular edema (DME) remains a major cause of vision loss in patients with diabetes mellitus (types 1 and 2). It is a multifactorial disease, largely caused by a change in permeability across the blood–retinal barrier (BRB) as a result of hyperglycemia.1 Leakage of fluid across the compromised BRB then causes macular edema.2
Factors influencing treatment selection Several surgical and pharmacologic options are available for patients with DME. Understanding the underlying pathophysiology can influence treatment decisions; for example, clinical evidence suggests that during early-stage disease, vascular endothelial growth factor (VEGF) is primarily responsible for retinal changes; anti-VEGF therapy is, therefore, effective at this stage. In the longer term, however, chronic microglial activation, resulting from retinal damage, leads to cytokine production by retinal cells and the initiation of inflammatory cascades, and at this later stage, corticosteroids are most effective.3 In addition to understanding the pathophysiology, the phenotypic differentiation of DME may be useful in planning treatment, as well as for improving the visual and anatomical prognosis.4 Structural and functional retinal imaging techniques are used to provide information about the individual disease characteristics in each patient. Commonly used techniques include optical coherence tomography (OCT), fluorescein angiography (FA), fundus photography, microperimetry, and fundus autofluorescence (Table 1).
Table 1. Imaging techniques used in DME.

Different phenotypes of DME Diabetic macular edema is clinically classified as diffuse, local or both, while four categories of DME have been established based on OCT retinal imaging:1
- diffuse DME or diffuse retinal thickening (DRT)
- cystoid macular edema (CME)
- serous sensory detachment DME (SRD)
- vitreomacular interface abnormalities (VMIA).

Diffuse DME or DRT Diffuse DME or DRT is characterized by retinal thickening, weakening of the light reflection and irregular low reflex zone, often with cystoid changes around the fovea and often without clinically visible capillary non-perfusion.1,9 It can be visualized using FA and OCT.6 Diffuse edema in large areas of the retinal capillary bed is caused by leakage from microaneurysms, retinal capillaries and arterioles, and is the result of a breakdown of the BRB.6,9,10 Patients with diffuse leakage in the fovea have a relatively poor prognosis. Before the introduction of anti-VEGF agents, modified grid macular laser photocoagulation was the preferred, but often ineffective, treatment.9 In contrast, focal leakage causes local, discrete swelling of the retina.2,9 It is associated with hard exudate rings (yellow deposits of lipid and protein), caused by leakage from microaneurysms and can be visualized using FA and fundus photography (Figure 1).2,3,6,10 The swelling is likely caused by loss of the BRB in the retinal capillaries near areas of ischemia, and is usually responsive to focal laser photocoagulation.9
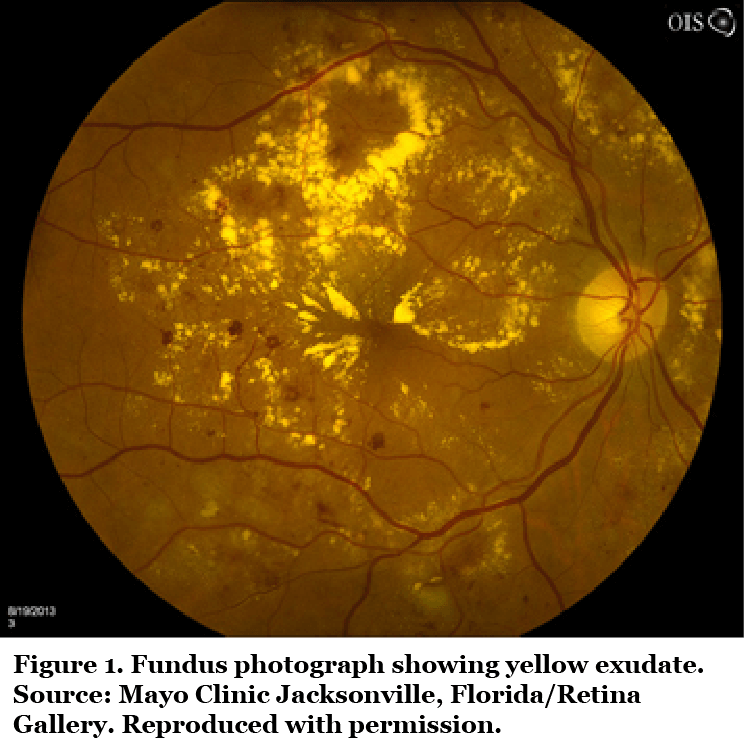
Cystoid macular edema In CME, dark cystoid spaces consisting of fluid that appears clear ophthalmoscopically may be detected in the macular area by OCT (Figure 2) or FA. These cysts seem to be areas of the retina from which cells have been displaced.1,10
Figure 2. Optical coherence tomography (OCT) image showing CME. Source: Retina Gallery. Reproduced with permission.
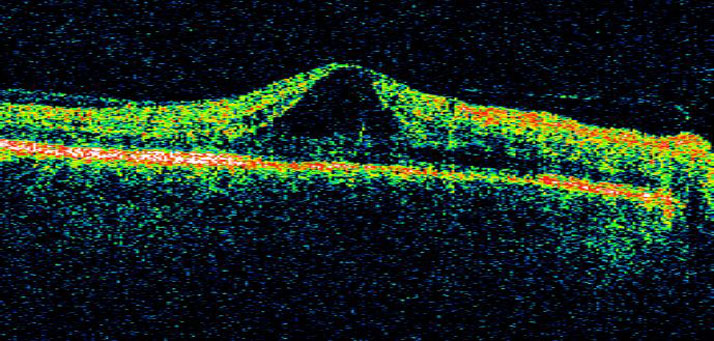
Good vision may be restored when the intraretinal fluid is removed.10 Several clinical trials have confirmed that in CME, pars plana vitrectomy surgery effectively reduces macular edema via several mechanisms, increasing oxygen supply to the retina and alleviating the ischemia.
Serous sensory detachment DME Serous sensory detachment is characterized by neuronal sensory or pigment epithelial detachment, and can be visualized by OCT (Figure 3).1 Neurosensory retinal detachment can be defined as subfoveal fluid accumulation under the detached retina, with or without overlying foveal thickening.11
Figure 3. Optical coherence tomography (OCT) image showing SRD. Source: Retina Gallery. Reproduced with permission.
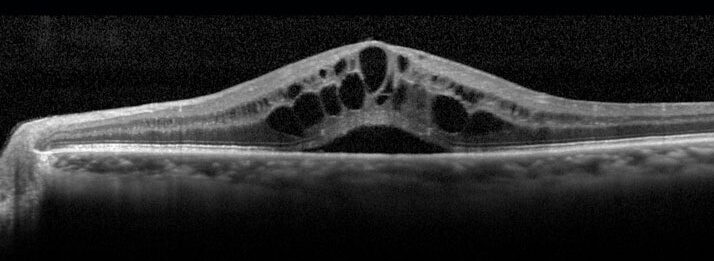
The presence of neurosensory retinal detachment adversely affects the prognosis of DME. The effectiveness of laser treatment of the macula in SRD is limited, and the presence of SRD in DME associated with subretinal exudation is associated with poor visual prognosis after vitrectomy.11
Vitreomacular interface abnormalities The VMIA category of DME is characterized by incomplete or complete posterior vitreous detachment,1 which can result in:
- vitreomacular adhesion (VMA)
- vitreomacular traction (VMT)
- epiretinal membrane
- lamellar hole
- full-thickness macular hole.12
Figure 4. Optical coherence tomography (OCT) image showing VMT. Source: Retina Gallery. Reproduced with permission.
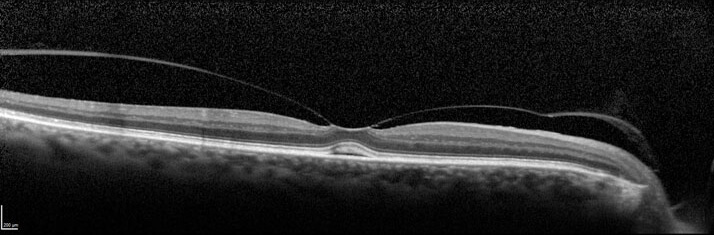
Monitoring the vitreomacular interface using OCT allows clinicians to identify VMA progression to VMT, detect mild epiretinal membranes, distinguish between lamellar holes and full-thickness macular holes, and measure the size of macular holes.12 This enables clinicians to select appropriate candidates for pharmacologic treatments or surgery. Vitrectomy can be useful in DME if there is evidence of VMT. In one published study, vitrectomy resulted in a 61–73 per cent resolution of edema in patients with DME without overt vitreoretinal interface abnormalities.10
Concluding remarks The use of an integrated structural and functional retinal imaging approach may lead to the identification of different DME phenotypes, which may allow prospective evaluation of treatment prognosis,13 and thereby influence treatment decisions.
Look out for DME content developed by Alimera Sciences on this website throughout 2015 and 2016. We hope it supports your knowledge of DME, and if you would like to contribute material for publication, please send your materials to dmecontenthub@hayward.co.uk, we’d be very pleased to consider your contributions.

REFERENCES
- X Zhang et al., “Diabetic macular edema: new concepts in patho-physiology and treatment”, Cell Biosci, 4, 27 (2014). PMID: 24955234. Available at: www.cellandbioscience.com/content/4/1/27. Accessed October 26, 2015.
- N Bhagat et al., “Diabetic macular edema: pathogenesis and treatment”, Surv Ophthalmol, 54, 1–32 (2009). PMID: 19171208.
- AJ Augustin et al., “Management of diabetic macular oedema – challenges and solutions along the patient journey”, Eur Ophthal Rev, 7, 115–120 (2013). Available at: www.touchophthalmology.com/articles/management-diabetic-macular-oedema-challenges-and-solutions-along-patient-journey-0. Accessed October 26, 2015.
- B PinaMarin et al., “Navina trial: diabetic macular edema treated with ranibizumab alone or in combination with other treatments, based on an individualized treatment scheme (phenotype differentiation)”, 15th EURETINA Congress, Nice, France (2015). Available at: euretina.org/nice2015/programme/free-papers-details.asp. Accessed October 26, 2015.
- D Boyer, “Vitreomacular adhesion: pathology and treatment”, Retina Today, (2013). Available at: retinatoday.com/2013/03/vitreomacular-adhesion-pathology-and-treatment/. Accessed October 26, 2015.
- E Mavrikakis et al., “Macular edema in diabetes”. Available at: emedicine.medscape.com/article/1224138overview. Accessed October 26, 2015.
- E Midena, S Vujosevic, “Microperimetry in diabetic retinopathy”, Saudi J Ophthalmol, 25, 131–135 (2011). PMID: 23960914.
- S Vujosevic et al., “Diabetic macular edema: fundus autofluorescence and functional correlations”, Invest Ophthalmol Vis Sci, 52, 442–448 (2011). PMID: 20720226.
- I Klaassen et al., “Molecular basis of the inner blood-retinal barrier and its breakdown in diabetic macular edema and other pathological conditions”, Prog Retin Eye Res, 34, 19–48 (2013). PMID: 23416119.
- T Rotsos, M Moschos, “Cystoid macular edema”, Clin Ophthalmol, 2, 919–930 (2008). PMID: 19668445.
- A Gupta et al., “Association of systemic and ocular risk factors with neurosensory retinal detachment in diabetic macular edema: a case–control study”, BMC Ophthalmol, 14, 47 (2014). PMID: 24716846. Available at: www.ncbi.nlm.nih.gov/pmc/articles/PMC3984633/ Accessed October 26, 2015.
- NW Muakkassa, MC Liang, “A close look at ocriplasmin for VMI disease. The use of this recombinant protease requires an appreciation for the history of VMI”, Ophthalmol Management, 18, 37–40 (2014). Available at: www.ophthalmologymanagement.com/articleviewer.aspx?articleID=111399. Accessed October 26, 2015.
- E Midena et al., “Diabetic macular edema phenotypes: the imaging perspective”, Invest Ophthalmol Vis Sci, 50, 3297 (2009).
Founded in 2003, Alimera Sciences researches and develops innovative vision-improving treatments for chronic retinal diseases, such as diabetic macular edema (DME), dry age-related macular degeneration (AMD), and retinal vein occlusion. In 2015, Alimera Sciences partnered with The Ophthalmologist to facilitate the publication of independently created educational content surrounding DME, a serious retinal complication associated with diabetes, which is increasing in incidence with the increasing prevalence of diabetes worldwide. Published content will include articles ranging from basic science and disease processes to overviews of clinical data, different surgical procedures, comparisons of treatment options, and practical advice for managing diabetic patients. With a commitment to honesty, integrity, responsibility, candor, and trust, Alimera Sciences intend to provide educationally focused content to healthcare professionals across a wide range of topics in DME in order to both increase disease awareness and understanding, and to help improve patient outcomes. UK-ILV-MMM-0355 Date of preparation: August 2015 enquiries@alimerasciences.com

