
Educational content provided by Alimera Sciences.
Diabetic macular edema (DME) is the main cause of vision loss associated with diabetic retinopathy (DR),1 and is characterized by vascular leakage, tissue edema and the deposition of hard exudates in the central retina.2 The main treatment modalities currently in use for DME include laser photocoagulation, intravitreal anti-vascular endothelial growth factors (anti-VEGFs), and intravitreal corticosteroids or corticosteroid implants.3,4 Although focal and/or grid laser photocoagulation was long seen as the mainstay of treatment for patients with DME, it only stabilizes vision, with many patients suffering vision loss despite having received laser therapy.5 More recently, the use of anti-VEGFs or corticosteroids has revolutionized the management of DME. Intravitreal anti-VEGF injections have been shown to significantly improve vision and restore retinal anatomy in many patients, and intravitreal corticosteroid injections have been shown to reduce DME, thus improving vision.6,7 Intravitreal injection of non-steroidal anti-inflammatory drugs (NSAIDs) has shown promising results, as have sustained release corticosteroid intravitreal implants, which have demonstrated significant improvements in visual acuity, reducing central macular thickness (CMT) in DME patients.6,8,9,10,11,12 When choosing a treatment, clinicians and patients need to consider the impact that treatment has on patient quality of life (QoL). Despite demonstrating efficacy in DME, intravitreal injection of anti-VEGFs or corticosteroids can be associated with a significant treatment burden. This article highlights the effects that DME and, more specifically, its treatment with intravitreal injection therapy can have on patients’ QoL.
Impact of DME and intravitreal injection therapy on patients’ QoL Diabetic macular edema and its treatment can both directly and indirectly affect patients’ QoL. Several studies have tried to quantify their impact. One of these studies, a survey—supported by Allergan Ltd.—of 131 patients with retinal disease (66 percent with DME and 34 percent with retinal vein occlusion), aimed to understand the treatment burden, treatment-related anxiety and practical issues in patients receiving intravitreal injection therapy. A number of patient QoL-related factors were identified as being affected by the treatment burden associated with intravitreal injection therapy (Figure 1).13
Figure 1. Treatment burden associated with intravitreal injection therapy.13
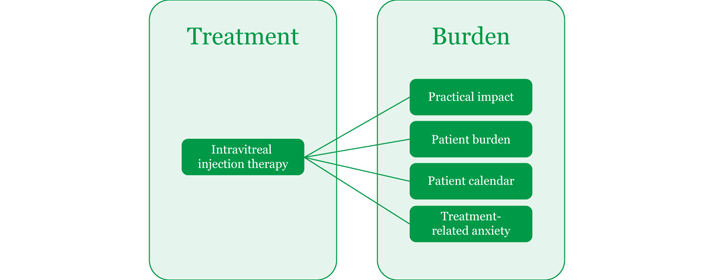
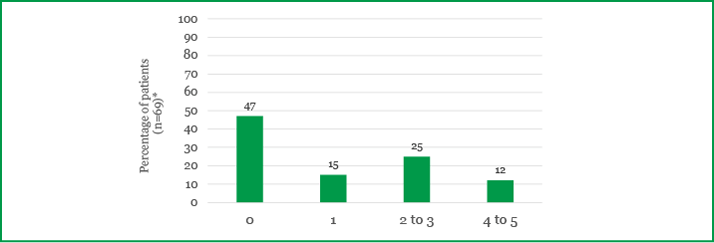
Practical impact Treatment of DME can result in time-related costs for both patients and carers. The survey found that patients spent a total of four hours 27 minutes on average per appointment, including travel time. In addition, just over half of the working patients had at least one day off work per injection, including recovery time (Figure 2). Furthermore, a large proportion of patients (71 percent) needed support from a carer for each injection. On average, each carer was perceived to spend 5.3 hours with the patient for each injection, and many of those (59 percent) needed time off from work to support patients.13
Figure 2. Total time off work per injection, including recovery time. *Only includes patients in employment.13
Patient burden Diabetes patients who suffer from multiple comorbid chronic diseases are often burdened with frequent hospital and healthcare visits. This could be exacerbated by the need for repeated intravitreal injection therapy during DME treatment. The survey found that patients with DME spent a total of 20 hours every six months attending appointments.13 Patient calendar According to the survey, over a six-month period, more than half of the patients with DME will have approximately 19 appointments with a number of healthcare professionals, as well as additional appointments for comorbid conditions. This significant appointment burden not only has an impact on patients’ time, but could also make it difficult for patients to attend every appointment.13 DME- and DME treatment-related anxiety Patients with retinal disease can also suffer from disease- and treatment-related anxiety. This is clear, for example, in a Canadian study (C-Reality), where 145 patients with DME were assessed at baseline, and at Months 3 and 6. Health-related QoL, including mental health, was assessed using the National Eye Institute Visual Functioning Questionnaire-25 item and EuroQol 5 dimensions. The study concluded that, as visual acuity worsened in patients with DME, mental-health scores can also worsen.14 Treatment of DME can also impact on the mental health of patients, as shown by Sobha Sivaprasad (Consultant Ophthalmologist at Moorfields) and Sesen Oyetunde (International Scientific Advisor at Allergan Ltd.) at the 6th World Congress on Controversies in Ophthalmology, who found that a high proportion of patients (75 percent) reported experiencing anxiety with their most recent intravitreal injection, and just over half felt anxious for two or more days prior to treatment. Table 1 shows the effect that intravitreal injection therapy had on patient anxiety in this survey.13
Table 1. Effect of intravitreal injection therapy on patient anxiety.13

Improvement to procedure Based on the data presented by Sivaprasad and Oyetunde, the improvement to treatment procedure most desired by patients is to have fewer intravitreal injections with the same visual results. Many patients would also like to attend fewer appointments for their treatment, while still achieving the same outcome.13
Conclusion Treatment burden, including co-morbidities, DME- and treatment-related anxiety, QoL and practical issues regarding travel time, absence from work, treatment adherence, and appointment attendance, should be carefully considered when choosing the adequate treatment option for patients with DME. Furthermore, it is important that clinicians also consider treatment burden on carers, as it highly likely that DME patients will require a carer to attend appointments with them, as well as help and support them during and after treatment. With intravitreal injection therapy, patients are generally initiated on a monthly injection course, which results in high anxiety burden and impact on QoL.13

Look out for DME content developed by Alimera Sciences on this website throughout 2015. We hope it supports your knowledge of DME, and if you would like to contribute material for publication, please send your materials to dmecontenthub@hayward.co.uk, we’d be very pleased to consider your contributions.
REFERENCES
- R Adelman et al., “Strategy for the management of diabetic macular edema: the European Vitreo-Retinal Society macular edema study”, Biomed Res Int, 2015, 1–9 (2015). PMID: 25695062 (http://www.hindawi.com/journals/bmri/2015/352487/).
- DS Boyer et al., “Anti-vascular endothelial growth factor therapy for diabetic macular edema”, Ther Adv Endocrinol Metab, 4, 151–69 (2013). PMID: 24324855 (http://tae.sagepub.com/content/4/6/151).
- MM Nentwich, MW Ulbig, “Diabetic retinopathy—ocular complications of diabetes mellitus”, World J Diabetes, 6, 489–499 (2015). PMID: 25897358 (http://www.wjgnet.com/1948-9358/full/v6/i3/489.htm).
- Diabetes UK, “Diabetic Retinopathy Treatment”. Available from: http://www.diabetes.co.uk/diabetes-complications/retinopathy-treatment.html. Accessed September 2015.
- F Bandello et al., “New approaches for the treatment of diabetic macular oedema: recommendations by an expert panel”, Eye, 26, 485–493 (2012). PMID: 22241014 (http://www.nature.com/eye/journal/v26/n4/full/eye2011337a.html).
- C Mathew et al., “Updates in the management of diabetic macular edema”, J Diabetes Res, 2015, 1–9 (2015). PMID: 25984537 (http://www.hindawi.com/journals/jdr/2015/794036/).
- QD Nguyen et al., on behalf of the RISE and RIDE Research Group, “Ranibizumab for diabetic macular edema. Results from 2 phase III randomized trials: RISE and RIDE”, Ophthalmology, 119, 789–801 (2012). PMID: 22330964.
- PA Pearson et al., “Fluocinolone acetonide intravitreal implant for diabetic macular edema: a 3-year multicenter, randomized, controlled clinical trial”, Ophthalmology, 118(8), 1580–1587 (2011). PMID: 21813090.
- PA Campochiaro et al., “Sustained delivery fluocinolone acetonide vitreous inserts provide benefit for at least 3 years in patients with diabetic macular edema.” Ophthalmology, 119(10), 2125–2132 (2012). PMID: 22727177.
- DG Callanan et al., “Dexamethasone intravitreal implant in combination with laser photocoagulation for the treatment of diffuse diabetic macular edema”, Ophthalmology, 120(9), 1843–1851 (2013). PMID: 23706947.
- Diabetic Retinopathy Clinical Research Network, “Three-year follow-up of a randomized trial comparing focal/grid photocoagulation and intravitreal triamcinolone for diabetic macular edema”, Arch Ophthalmol, 127(3), 245–251 (2009). PMID: 19273785. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2754047/pdf/nihms-141059.pdf
- Diabetic Retinopathy Clinical Research Network, “A randomized trial comparing intravitreal triamcinolone acetonide and focal/grid photocoagulation for diabetic macular edema”, Ophthalmology, 115, 1447–1459 (2008). PMID: 18662829.
- S Sivaprasad, S Oyetunde, “Impact of injection therapy for retinal patients with DME or RVO”, poster presented at the 6th World Congress on Controversies in Ophthalmology (COPHy), Sorrento, Italy; 26–29 March, 2015.
- JR Gonder et al., “Costs and quality of life in diabetic macular edema: Canadian Burden of Diabetic Macular Edema Observational Study (C-REALITY)”, Journal of Ophthalmology, 2014, 1–9 (2014). PMID: 24795818 (http://dx.doi.org/10.1155/2014/939315).
Founded in 2003, Alimera Sciences researches and develops innovative vision-improving treatments for chronic retinal diseases, such as diabetic macular edema (DME), dry age-related macular degeneration (AMD), and retinal vein occlusion. In 2015, Alimera Sciences partnered with The Ophthalmologist to facilitate the publication of independently created educational content surrounding DME, a serious retinal complication associated with diabetes, which is increasing in incidence with the increasing prevalence of diabetes worldwide. Published content will include articles ranging from basic science and disease processes to overviews of clinical data, different surgical procedures, comparisons of treatment options, and practical advice for managing diabetic patients. With a commitment to honesty, integrity, responsibility, candor, and trust, Alimera Sciences intend to provide educationally focused content to healthcare professionals across a wide range of topics in DME in order to both increase disease awareness and understanding, and to help improve patient outcomes. UK-ILV-MMM-0355 Date of preparation: August 2015 enquiries@alimerasciences.com

