
Advertisement feature; authors report for the first time outcomes with fluocinolone acetonide intravitreal implant versus an anti-VEGF in chronic diabetic macular edema
Funded by Alimera Sciences. Contains promotional information. Please click here to view
the prescribing information and adverse event reporting information.
A recent case study, published in Ophthalmology and Therapy, describes the outcomes over
more than one year following administration of the ILUVIEN® (fluocinolone acetonide)
intravitreal implant at 0.2 μg per day (licensed dose) in a patient with chronic diabetic
macular edema (DME) not responding to laser photocoagulation or anti-vascular endothelial
growth factor (anti-VEGF) therapy. The authors—Thomas Bertelmann and Stephan Schulze,
from Philipps University, Marburg, Germany—chose initially to treat only the patient’s left
eye with the FAc intravitreal implant, while anti-VEGF injections were continued in the right
eye. This study provides the first direct comparison between anti-VEGF in one eye and an
FAc intravitreal implant in the fellow eye. Furthermore, results provide an insight into the
relative efficacy of the FAc intravitreal implant over a 12-month period, in chronic DME
unresponsive to anti-VEGF therapy.1
Although anti-VEGF agents have, in many cases, become the preferred treatment for DME, it
has been reported that as many as 42% of patients do not respond adequately to this
therapy.1,2 In such cases, intravitreal corticosteroids may provide an efficacious alternative.1
The study by Bertelmann and Schulze focuses on a single 30-year-old patient with type 1
diabetes mellitus who was diagnosed with DME 20 years ago. Laser photocoagulation had
been administered in both eyes and this was followed, since 2011, by 15 injections of the
anti-VEGF ranibizumab in the right eye and 12 injections in the left eye.1
In August 2013, following completion of the initial treatment strategies, the FAc intravitreal
implant was administered in the patient’s left eye. Within one month, central macular
thickness (CMT) decreased from 642 μm at baseline to 278 μm. Marked reductions in CMT
remained evident over 13 months of follow-up. Likewise, best corrected visual acuity (BCVA)
improved from 0.3 at baseline to 0.5 at month one; an improvement that was sustained for
nine months. After 13 months, BCVA decreased to 0.4, owing to cataract formation, but
returned to 0.5 one day after phacoemulsification.1
While the left eye was treated with the FAc intravitreal implant, the right eye received three
additional injections of ranibizumab (a total of 18 injections). Although CMT in the right eye
decreased over seven months—by 166 μm from a baseline of 639 μm—with continued
ranibizmab treatment, this contrasts to the drop of 364 μm from a baseline of 642 μm
observed after one month post FAc intravitreal implant in the left eye. Furthermore, with
ranibizumab treatment, the macula in the right eye remained swollen and wet. Following
administration of the FAc intravitreal implant to the right eye, a further drop in CMT was
seen—of 210 μm after one month to 263 μm—with the level increasing slightly to 334 μm
at month six.1
BCVA improved from 0.5 at baseline to 0.6 at one month post FAc intravitreal implant, which
further increased to 0.7 at month six (Figure 1). These observations suggest that for this
patient, FAc was an improvement on continued anti-VEGF therapy.1
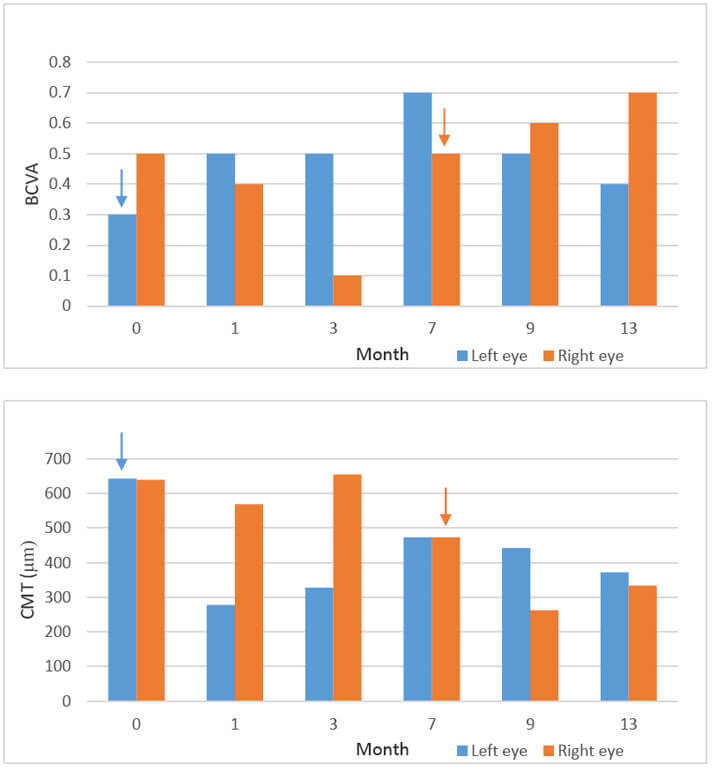
Figure 1. Changes in BCVA and CMT before and after the FAc implant. Graphs show BCVA
and CMT in left and right eyes. Arrows indicate when the FAc intravitreal implant was first
inserted. ETDRS; Early treatment Diabetic Retinopathy Study. Adapted from Bertelmann and
Schulze. 2014.1
The patient, who had left employment as a window cleaner on high-rise buildings as a result
of chronic DME, reported that he had been able to begin working again following treatment
with the FAc intravitreal implant. This is most likely a result of the improvement in BCVA to
0.5, or 6/12, with the corticosteroid, which is also the minimum requirement for maintaining
a driving license in the UK.1,3
The adverse event associated with the FAc intravitreal implant in this study was an increase
in intraocular pressure (IOP) in the left eye, from 20 mmHg at month three to 32 mmHg at
month seven. Treatment with 10 mg brinzolamide/5 mg timolol eye drops reduced this to
16mmHg. At 13 months left and right eye IOP were at 21 and 18mmHg respectively. No
further adverse events were reported at the time this case study was accepted for
publication, January 2015.1
The observations from this single-patient case study suggest that the FAc intravitreal
implant could be effective in the management of chronic DME in a real-world clinical
setting, and that this corticosteroid should be considered in chronic DME that is
unresponsive to anti-VEGF and/or laser treatment. In addition, the comparable response to
the FAc intravitreal implant between the left and right eyes in this patient indicates that the
outcome in one eye with a particular treatment may be an indication of the subsequent
response in the other eye with the same treatment.
Look out for DME content developed by Alimera Sciences on this website throughout 2015.
We hope it supports your knowledge of DME and ILUVIEN, and if you would like to
contribute material for publication, please send your materials to
dmecontenthub@hayward.co.uk, we’d be very pleased to consider your contributions.
REFERENCES 1. Bertelmann T and Schulze S. Long-Term Follow-Up of Patient with Diabetic Macular Edema Receiving Fluocinolone Acetonide Intravitreal Implant. Ophthal Ther, Epub ahead of print (2015). PMID: 25672501 (http://dx.doi.org/10.1007/s40123-015-0028-0) 2, Brown DM, Nguyen QD, Marcus DM, et al. Longterm outcomes of ranibizumab therapy for diabetic macular edema: the 36-month results from two Ophthalmol Ther phase III trials: RISE and RIDE. Ophthalmology. (2013) 120:2013–22 3. Driving eyesight rules. Gov.uk. Available at www.gov.uk/driving-eyesight-rules (Accessed May 2015)
UK-ILV-MMM-0207
Date of preparation: May 2015
Founded in 2003, Alimera Sciences researches and develops innovative vision-improving treatments for chronic retinal disease. Alimera Sciences has developed and licensed ILUVIEN®, an intravitreal implant of 190 micrograms fluocinolone acetonide, for the treatment of vision impairment associated with chronic diabetic macular edema (DME), considered insufficiently responsive to available therapies.a ILUVIEN is the first DME treatment to deliver up to 36 months of continuous, low-dose corticosteroid by single injection.b In 2015, Alimera Sciences has partnered with The Ophthalmologist to facilitate the publication of independently created content on ILUVIEN and DME. Content will range from conference reports, case studies, and literature reviews to video interviews, presentations, and practical information surrounding the use and benefits of ILUVIEN. The word Alimera derives loosely from the Greek, to mean “day of truth”.c With a commitment to honesty, integrity, responsibility, candor, and trust, Alimera Sciences intend that this promotional information accurately and fairly represents the current state of knowledge of ILUVIEN and DME, and is useful to all healthcare professionals involved in DME and its treatment. a. ILUVIEN SPC. 2013 Available at: www.medicines.org.uk/emc/medicine/27636 (Accessed March 2015) b. Alimera Sciences. Available at www.alimerasciences.com (Accessed March 2015) c. Retina Today. 2011. Available at: http://retinatoday.com/pdfs/0111RT_Wall.Street.pdf (Accessed March 2015) UK-ILV-MMM-0167 Date of preparation: March 2015 www.alimerasciences.com enquiries@alimerasciences.com

