
Advertisement feature; case study author explains why switch to a fluocinolone acetonide intravitreal implant should not be delayed in chronic diabetic macular edema patients not responding to anti-VEGFs
Funded by Alimera Sciences. Contains promotional information. Please click here to view the prescribing information and adverse event reporting information.
A single-patient case study (The Treatment of Diabetic Macular Oedema with ILUVIEN® Intravitreal Implant Following Prior Anti-VEGF Therapy) of the treatment of chronic diabetic macular edema (DME) showed that an ILUVIEN® (fluocinolone acetonide) intravitreal implant (Alimera Sciences) reduced the treatment burden in a non-responder to anti-vascular endothelial growth factor (anti-VEGF) therapy, while improving their visual outcomes and macular thickness, with no evidence of intraocular hypertension (IOH), according to Mr Fahd Quhill.1,2
Fahd Quhill is a Consultant Ophthalmologist at the Royal Hallamshire Hospital, Sheffield, UK, having previously completed several medical retinal fellowships, in Birmingham, Wolverhampton and at Moorfields Eye Hospital, London, UK. He currently serves in a large tertiary referral service for macular diseases, with specialist interests in diabetic retinopathy, retinal vascular diseases, uveitis, ocular oncology, electrophysiology, and retinal dystrophy.

The case study describes a 68-year-old female with type 2 diabetes and DME. The patient’s DME was initially treated with laser and intravitreal injections of triamcinolone acetonide, followed by combination therapy with the small interfering-RNA agent PF-04523655 (siRNA) and ranibizumab or siRNA alone as part of the MATISSE trial. Subsequently, she received further injections of ranibizumab. After a total of 11 injections, little improvement in macular edema or visual function was observed.1,2
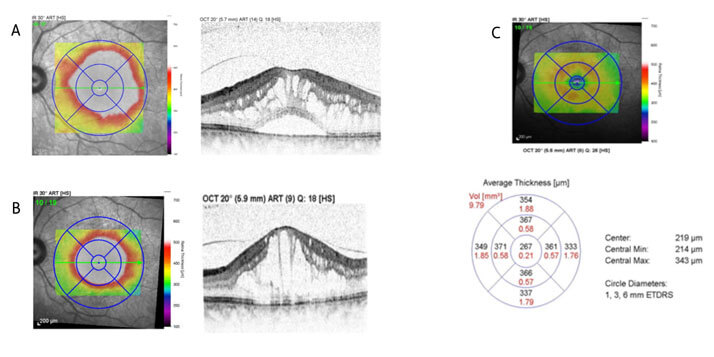
Following the completion of the initial strategies, treatment with an ILUVIEN implant was initiated. Within 7 days of a single injection, macular thickness decreased by more than 500 μm and after a further 14 days, visual acuity improved by 15 letters.1,2 These improvements were sustained after 12 months. Throughout this period, no IOH was observed and no re-treatment with ILUVIEN or administration of another DME treatment method was required.1,2
Improvements 1 week after injection:
- Left eye visual acuity improved to 6/18
- Central macular thickness decreased from 774 μm to 267 μm
- Decimal vision improved from 0.25 to 0.33
- Vision improved to 0.50 (6/12); that is, a 15-letter gain maintained at 6 months
Figure 1: Optical coherence tomography scan showing foveal thickness at baseline prior to anti-VEGF treatment (A), following 11 anti-VEGF injections (B), and one week post-treatment with ILUVIEN (C), reproduced with permission from Fahd Quhill. 2015
This case demonstrates the importance of earlier identification of non-responders to anti-VEGF treatment and initiating another treatment method, such as ILUVIEN as rapidly as possible. According to Fahd Quhill, “[Non-responders] shouldn’t be subjected to continued anti-VEGF injections if clearly they are not getting the improved vision and resolution of macular edema that one would expect with anti-VEGF treatment. The watch-and-wait approach is counterproductive because by the time you do make the [treatment] switch, you might not be able to restore sight back up to satisfactory levels, even if macular edema has been resolved.” It is equally important to recognize that there are two types of DME patients: one who is responsive to anti-VEGF treatment and a second who does not respond. It is down to the treating ophthalmologist to ensure that patients in the latter group are quickly identified and offered an alternative treatment. As reinforced by Mr Quhill, “The key is not to delay the switch to [ILUVIEN] in these patients.”2
So, which patients most benefit from a switch to the ILUVIEN implant? Mr Quhill suggests, “Anti-VEGF non-responders – defined in my clinic as someone who has not responded after three or four injections [with] at least a 120–130 μm reduction in macular edema or at least a 5- to 6-letter gain in vision after three to four injections – are the kinds of patients I would want to consider switching.” Such patients are likely to have chronic diabetic maculopathies, where an inflammatory pathway, in addition to VEGF, is driving the macular edema. It is in such cases that ILUVIEN has demonstrated the greatest efficacy. Patients who have previously responded to corticosteroids or those who have difficulty coping with the multi-injection regimen associated with anti-VEGF therapy may also be candidates for a switch to the ILUVIEN intravitreal implant. Mr Quhill feels that, “A patient who is fed up of having lots of anti-VEGF injections, who can’t cope with the degree of treatment, may consider ILUVIEN to give them a longer-term sustained delivery.”2
Switching to ILUVIEN had an enormous impact on this patient, as noted at the 12-month follow-up, while benefits of ILUVIEN should be seen for up to three years. In Mr Quhill’s experience, “If it works, it means [patients] do not have to be given any other intravitreal therapy for at least 12 months. In other cases, it may mean patients only need additional laser treatment top-ups. For patients who are non-responsive to anti-VEGFs, this [ILUVIEN implant] is able to offer the opportunity to make gains in vision and get them back to driving: back to doing the things that they need vision to do.” Not considering a change in therapy is, according to Mr Quhill, to “Miss out on the possibility of restoring sight to levels of 6/12, which is an important cut-off for most [opportunities to improve] patient’s lifestyles.” Additionally, Mr Quhill feels that by delaying the switch from anti-VEGF therapy to ILUVIEN, patients are potentially denied the opportunity for visual rehabilitation.2
Key case study findings
In a single-patient case study, switching to a fluocinolone acetonide intravitreal implant following no response to anti-VEGF therapy led to a rapid and sustained improvement in visual outcomes and macular thickness, with no evidence of IOH.1 Patients who have shown insufficient responsiveness to therapies, such as anti-VEGF, are candidates for earlier intervention with a fluocinolone acetonide intravitreal implant, which can reduce reliance on anti-VEGF agents and potentially lower the associated treatment burden. There is a core group of DME patients who are anti-VEGF resistant. These patients should be quickly identified and a switch to fluocinolone acetonide intravitreal implants should not be delayed.
Look out for DME content developed by Alimera Sciences on this website throughout 2015. We hope it supports your knowledge of DME and ILUVIEN. If you would like to contribute material for publication, please send your materials to dmecontenthub@hayward.co.uk. We’d be very pleased to consider your contributions.
REFERENCES 1. F Quhill, “The Treatment of Diabetic Macular Oedema with ILUVIEN® Intravitreal Implant Following Prior Anti-VEGF Therapy – Case Study”, Eur Opth Rev, 8, 140–144 (2014). Available at: www.touchophthalmology.com/articles/treatment-diabetic-macular-oedema-iluvien-intravitreal-implant-following-prior-anti-vegf (Accessed May 2015). 2. Interview with Fahd Quhill, April 2015.
UK-ILV-MMM-0175 Date of preparation: May 2015
Founded in 2003, Alimera Sciences researches and develops innovative vision-improving treatments for chronic retinal disease. Alimera Sciences has developed and licensed ILUVIEN®, an intravitreal implant of 190 micrograms fluocinolone acetonide, for the treatment of vision impairment associated with chronic diabetic macular edema (DME), considered insufficiently responsive to available therapies.a ILUVIEN is the first DME treatment to deliver up to 36 months of continuous, low-dose corticosteroid by single injection.b In 2015, Alimera Sciences has partnered with The Ophthalmologist to facilitate the publication of independently created content on ILUVIEN and DME. Content will range from conference reports, case studies, and literature reviews to video interviews, presentations, and practical information surrounding the use and benefits of ILUVIEN. The word Alimera derives loosely from the Greek, to mean “day of truth”.c With a commitment to honesty, integrity, responsibility, candor, and trust, Alimera Sciences intend that this promotional information accurately and fairly represents the current state of knowledge of ILUVIEN and DME, and is useful to all healthcare professionals involved in DME and its treatment. a. ILUVIEN SPC. 2013 Available at: www.medicines.org.uk/emc/medicine/27636 (Accessed March 2015) b. Alimera Sciences. Available at www.alimerasciences.com (Accessed March 2015) c. Retina Today. 2011. Available at: http://retinatoday.com/pdfs/0111RT_Wall.Street.pdf (Accessed March 2015) UK-ILV-MMM-0167 Date of preparation: March 2015 www.alimerasciences.com enquiries@alimerasciences.com

