
Advertisement feature; Mr Nishal Patel discusses the benefits of the guidelines, and the advantages of combining cataract removal surgery with application of the fluocinolone acetonide intravitreal implant
Funded by Alimera Sciences. Contains promotional information. Please click here to view the prescribing information and adverse event reporting information.
In an interview for The Ophthalmologist, Mr Nishal Patel—Consultant Ophthalmologist and Medical Retina Specialist at East Kent Hospitals University NHS Foundation Trust, UK—discusses the National Institute for Health and Care Excellence (NICE) guidelines on the use of the ILUVIEN® (fluocinolone acetonide) intravitreal implant (Alimera Sciences) for chronic diabetic macular edema (DME), and the potential benefits of combining cataract-removal surgery with ILUVIEN implantation.1
In 2013, NICE recommended ILUVIEN as an option for treating chronic DME that is unresponsive to other therapies, in patients receiving treatment in an eye with a pseudophakic lens.2 This guidance is welcomed by both patients and physicians, alike. Individuals with chronic DME who have shown a poor clinical response to anti-vascular endothelial growth factor (anti-VEGF) injections may have an inflammatory component to their eye disease that does not respond to anti-VEGF therapy, despite multiple treatments. ILUVIEN has been shown to release fluocinolone acetonide in the management of chronic DME for up to three years after a single application.3 “Patients are happy,” explains Mr Patel. “They don’t need multiple injections.”
From the point of view of physicians, Mr Patel and colleagues have found ILUVIEN to be “a well-tolerated, effective treatment,” particularly in the context of trying to reduce the number of injections administered to patients in whom either laser or anti-VGEF injections have failed to manage their chronic DME.
Mr Patel also reports that ILUVIEN benefits from a relatively simple intravitreal administration. “It’s a very quick and easy procedure,” he states. “In terms of the device insertion to the eye, it can be done in a treatment room pretty similarly to the way that anti-VGEF treatment injections [are performed].” For practitioners with concerns over needle size or the complexity of the device, a theater or day surgical setting is recommended for the procedure. As physician confidence with the procedure improves, administration can take place in treatment rooms, in an outpatient setting.
The NICE guidelines do, however, restrict the use of ILUVIEN to pseudophakic eyes. Thus, the number of patients who can be offered treatment may be relatively small, but Mr Patel feels that this is a sensible approach. “I think the criteria are tight, but they are tight for a reason: because of the cost-effectiveness of the therapy and the quality outcomes necessary. If you look at the FAME study, where 81.7% of patients developed cataracts in phakic eyes,4 it makes sense to do the procedure in pseudophakic eyes, because then that will change your outcomes and you will start afresh—you don’t have to wait for cataracts to develop and then wait for the outcomes to get better after cataract surgery. This way, you start with an eye that has had its cataract removed and [can see] the definitive effect of ILUVIEN and its ability to treat the inflammatory component of diabetic eye disease.”
But how are the NICE guidelines being implemented in clinical practice? Mr Patel treats patients with pseudophakic DME initially with an anti-VEGF, as recommended by NICE. If there is a lack of improvement with anti-VEGFs, ILUVIEN is initiated. Mr Patel feels that the quicker the corticosteroid is introduced, the better: “Recognizing that inflammation has a role to play in diabetic eye disease is quite important, and the earlier you intervene to correct that, the better the results will be.”
Figure 1. Combined cataract removal and ILUVIEN insertion, surgery performed by Nishal Patel. Photograph reproduced with permission from Nishal Patel. 2015.
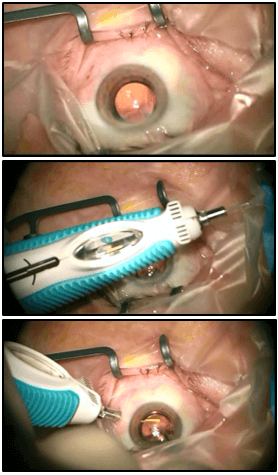
For patients with co-existing cataracts and persistent chronic DME despite multiple anti-VEGF treatments, Mr Patel recommends combined phacoemulsification intraocular lens (IOL) insertion, and ILUVIEN implantation (Figure 1). This approach requires only a single surgical visit and negates the need for intravitreal injection at a later date, as it is completed directly following cataract removal. Combined cataract removal and ILUVIEN implantation may have additional benefits. Indeed, Mr Patel feels that, “surgeons have to be aware that co-existing cataracts need to be considered. Treating the macular edema on the optical coherence tomography scan is one thing; but remembering that cataracts themselves are a more global cause of vision loss, and combining the surgery and treating the cataracts, and the macular edema with a single procedure may help to reduce the treatment burden to the NHS, and also treatment costs long term.”
The combined surgical approach may have other advantages for patients. Typically, acute inflammation occurs following cataract surgery, which can worsen DME and must be managed using topical corticosteroids. In vulnerable patients, intravitreal injections may be required to manage chronic inflammation. As such, a patient who has already received multiple intravitreal anti-VEGF injections, will require further injections. Mr Patel feels that, “if you imagine talking to a patient about the fact that they will require more injections and more drops after surgery, [this] will dissuade them from having cataract surgery. If you say, we can treat you with cataract surgery and we can inject the steroid at the same time, then that may treat the acute inflammation, and possibly reduce the need to have multiple injections later on; to the patient that is the more attractive option.”
So far, combined phacoemulsification IOL insertion and ILUVIEN implantation appear to be well-tolerated. It is important to bear in mind that corticosteroids, such as ILUVIEN, are associated with increases in intraocular pressure (IOP), so monitoring of patients is required after intravitreal injection. According to Mr Patel, however, “[patients] don’t mind coming in for scans. In my experience most do not have a significant rise in IOP, and those that do can be managed with appropriate therapies.” Furthermore, the combined approach reduces the risks associated with multiple ocular procedures, such as infection and other complications, which may occur following eye surgery.
Mr Patel does concede that it is early days for combined phacoemulsification IOL insertion and ILUVIEN implantation because, “at this stage … the number of surgeries performed as a combined surgery is so few, we need to gather that evidence to see if this might be a better way of managing patients long-term.” Mr Patel and his team plan to submit their data for combined surgery versus pseudophakic ILUVIEN implantation alone to the American Academy of Ophthalmology later this year.
A consultant ophthalmologist and medical retina specialist at East Kent Hospitals University NHS Foundation Trust, UK, Mr Patel considers ILUVIEN to be a well-tolerated, effective treatment, which can be quickly and easily implanted.

Look out for DME content developed by Alimera Sciences on this website throughout 2015. We hope it supports your knowledge of DME and ILUVIEN, and if you would like to contribute material for publication, please send your materials to dmecontenthub@hayward.co.uk. We’d be very happy to consider your contributions
REFERENCES 1. Interview with Nishal Patel, April 2015. 2. National Institute for Health and Care Excellence, “Fluocinolone acetonide intravitreal implant for treating chronic diabetic macular oedema after an inadequate response to prior therapy (rapid review of technology appraisal guidance 271). NICE technology appraisal guidance 301”, (2013). Available at www.nice.org.uk/guidance/ta301 (Accessed April 2015) 3. Alimera Sciences Limited, “ILUVIEN 190 micrograms intravitreal implant in applicator. Summary of Medical Product Characteristics”, 2014. 4. Campochiaro PA, Brown DM, Pearson A et al., “Sustained delivery fluocinolone acetonide vitreous inserts provide benefit for at least 3 years in patients with diabetic macular edema.”, Ophthalmology, 119, 2125–21324 (2012). PMID: 22727177. EUR-ILV-MMM-0073 Date of preparation: April 2015
Founded in 2003, Alimera Sciences researches and develops innovative vision-improving treatments for chronic retinal disease. Alimera Sciences has developed and licensed ILUVIEN®, an intravitreal implant of 190 micrograms fluocinolone acetonide, for the treatment of vision impairment associated with chronic diabetic macular edema (DME), considered insufficiently responsive to available therapies.a ILUVIEN is the first DME treatment to deliver up to 36 months of continuous, low-dose corticosteroid by single injection.b In 2015, Alimera Sciences has partnered with The Ophthalmologist to facilitate the publication of independently created content on ILUVIEN and DME. Content will range from conference reports, case studies, and literature reviews to video interviews, presentations, and practical information surrounding the use and benefits of ILUVIEN. The word Alimera derives loosely from the Greek, to mean “day of truth”.c With a commitment to honesty, integrity, responsibility, candor, and trust, Alimera Sciences intend that this promotional information accurately and fairly represents the current state of knowledge of ILUVIEN and DME, and is useful to all healthcare professionals involved in DME and its treatment. a. ILUVIEN SPC. 2013 Available at: www.medicines.org.uk/emc/medicine/27636 (Accessed March 2015) b. Alimera Sciences. Available at www.alimerasciences.com (Accessed March 2015) c. Retina Today. 2011. Available at: http://retinatoday.com/pdfs/0111RT_Wall.Street.pdf (Accessed March 2015) UK-ILV-MMM-0167 Date of preparation: March 2015 www.alimerasciences.com enquiries@alimerasciences.com

