
Advertisement feature; Marie Tsaloumas, Consultant Ophthalmic Surgeon at Queen Elizabeth Hospital, Birmingham, UK, reports effective use of ILUVIEN® (fluocinolone acetonide) intravitreal implant in a range of diabetic macular edema cases, including anti-VEGF unresponsive patients and those with vitrectomized eyes
Funded by Alimera Sciences. Contains promotional information. Please click here to view the prescribing information and adverse event reporting information.
Marie Tsaloumas was one of the presenters at Alimera Science’s diabetic macular edema (DME) Virtual Grand Round in November 2014.1 During her talk, entitled “ILUVIEN in diabetic macular oedema”, Miss Tsaloumas described her experience of using the fluocinolone acetonide intravitreal implant for the treatment of chronic DME.1 The presentation revealed important details regarding real-world clinical responses to the fluocinolone acetonide intravitreal implant among chronic DME patients, including those who had not responded adequately to treatment with anti-vascular endothelial growth factor (anti-VEGF) drugs or had undergone vitrectomy.
Some DME patients do not respond adequately to anti-VEGFs, and therefore need more treatment options. Suppression of inflammatory mediators is required for the successful management of chronic DME, which provides a rationale for the use of corticosteroids; corticosteriods act by reducing inflammation in the eye, as well as by decreasing VEGF levels and white blood cell infiltration.
Based on this, Miss Tsaloumas identified patients with chronic DME, who she felt would respond well to treatment with ILUVIEN. To date, 20 such patients have been selected and treated, Miss Tsaloumas presented data on a number of these patients, in each case, fluocinolone acetonide intravitreal implant was associated with rapid improvements in Snellen visual acuity (VA) and reductions in central retinal thickness (CRT), irrespective of prior response to anti-VEGFs.
One of the patients Miss Tsaloumas described was a 38-year-old with type 1 diabetes mellitus who had previously responded to treatment with the corticosteroid triamcinolone acetonide (TA), but had responded poorly to anti-VEGFs. The right eye, where VA was 6/18, was treated and, just one week after a single injection of ILUVIEN, VA improved to 6/12. The left eye, which had undergone vitrectomy and was much worse than the left, was treated at a later date. VA improved from 6/60 to 6/36 10 days post-operatively. This was accompanied by a large decrease in CRT in both eyes (Figure 1).
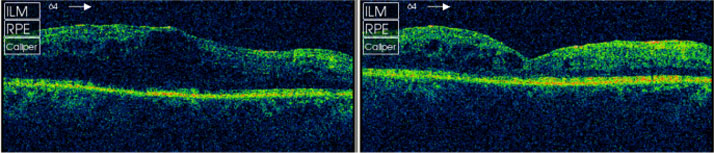
Figure 1. Central retinal thickness before (left) and one week after (right) a single injection of ILUVIEN in a vitrectomized patient with chronic DME unresponsive to anti-VEGFs.1 A similarly impressive response was reported in a 76-year-old patient with type 2 diabetes, who had a poor response to three injections of the anti-VEGF bevacizumab (Avastin®, Genentech/Roche) but some response to TA. Two months after a single injection of ILUVIEN, considerable improvements in the macula and reductions in CRT were noted (Figure 2).
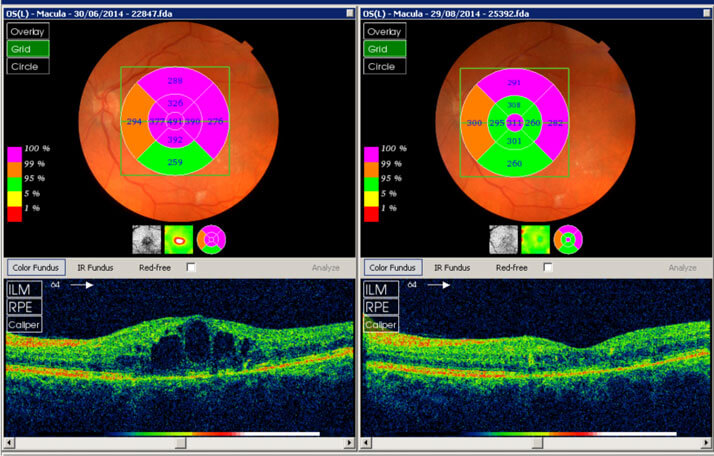
Figure 2. Macular imaging (top) and CRT (bottom) before treatment (left) and two months after treatment with ILUVIEN (right) in an elderly patient with chronic DME unresponsive to anti-VEGFs.1
Another patient of note had undergone renal pancreatic transplant - a procedure for the treatment of kidney failure related to type 1 diabetes in which both a kidney and the pancreas are replaced simultaneously.2 Despite this their diabetes, and associated DME, progressed and they required vitrectomy in both eyes. This patient was unresponsive to anti-VEGFs; however ILUVIEN led to rapid clinical and anatomical responses over a three month time period (Figure 3), which were being maintained after a further three months.
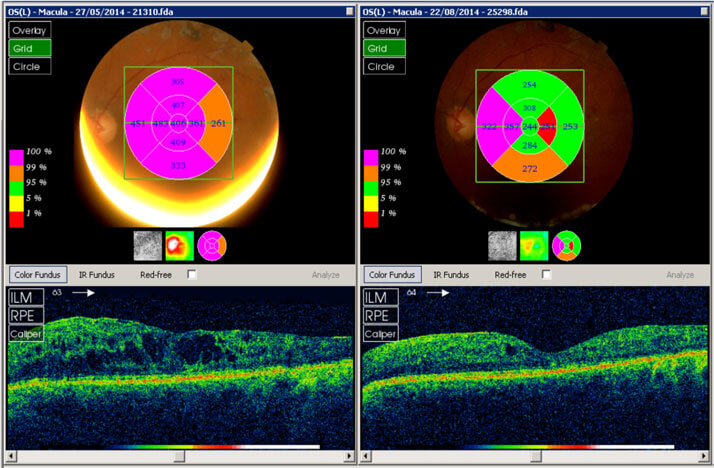
Figure 3. Macular imaging (top) and CRT (bottom) before treatment (left) and three months after treatment with ILUVIEN (right) in a renal pancreatic transplant patient with chronic DME unresponsive to anti-VEGFs.1
Miss Tsaloumas also highlighted how patients felt about the improvements in their VA. After treatment with ILUVIEN, patients reported marked improvements in their vision and one “couldn’t believe the clarity of their vision.” In many cases, the subjective improvement in VA was greater than the objective measurement. Based on these observations, Miss Tsaloumas suggested that a routine method of measuring changes in VA that is more sensitive than the Snellen chart – such as LogMAR or refractive VA assessment – is required. There may be additional benefit to including measurement of contrast sensitivity, reading ability, or reading speed to capture patients’ subjective improvements.
Miss Tsaloumas documented no unmanageable increases in intraocular pressure (IOP) in any of the patients during ILUVIEN treatment over a follow-up period of up to nine months. Monitoring of these patients is on-going. In most cases, IOP-lowering therapy was required after previous treatment with a different corticosteroid, but no extra treatment has been necessary after fluocinolone acetonide intravitreal implant administration (to date).
Miss Tsaloumas admitted that her findings raise some important questions regarding the use of ILUVIEN; for example, how should DME patients who may benefit from ILUVIEN treatment best be managed? Should cataract removal surgery and fluocinolone acetonide intravitreal implant injection take place at the same time or should the cataract be removed during separate surgery? Other important questions are how anti-VEGF unresponsiveness should best be defined and at what stage fluocinolone acetonide intravitreal implant should be introduced in place of these drugs. The answers to these questions should become apparent as more patients are treated with ILUVIEN.
Miss Tsaloumas, Consultant Ophthalmic Surgeon at Queen Elizabeth Hospital, Birmingham, UK, has a subspecialty interest in diabetic eye disease, age-related macular degeneration, and vascular diseases of the eye. She also runs regional and national multidisciplinary clinics dealing with inherited retinal conditions and their systemic manifestations.

Look out for DME content developed by Alimera Sciences on this website throughout 2015. We hope it supports your knowledge of DME and ILUVIEN, and if you would like to contribute material for publication, please send your materials to dmecontenthub@hayward.co.uk, we’d be very happy to consider your contributions. REFERENCES 1. M Tsaloumas, “ILUVIEN in diabetic macular oedema”, presented at the DME Virtual Grand Round, November 25, 2014, a virtual event organised by Alimera Sciences. Available at: events4healthcare.com/alimera_sciences/webinar25112014.html (Accessed May 2015) 2. National Kidney Foundation: Kidney-Pancreas Transplant Available at: www.kidney.org/atoz/content/kidpantx (Accessed May 2015) UK-ILV-MMM-0208 Date of preparation: May 2015
Founded in 2003, Alimera Sciences researches and develops innovative vision-improving treatments for chronic retinal disease. Alimera Sciences has developed and licensed ILUVIEN®, an intravitreal implant of 190 micrograms fluocinolone acetonide, for the treatment of vision impairment associated with chronic diabetic macular edema (DME), considered insufficiently responsive to available therapies.a ILUVIEN is the first DME treatment to deliver up to 36 months of continuous, low-dose corticosteroid by single injection.b In 2015, Alimera Sciences has partnered with The Ophthalmologist to facilitate the publication of independently created content on ILUVIEN and DME. Content will range from conference reports, case studies, and literature reviews to video interviews, presentations, and practical information surrounding the use and benefits of ILUVIEN. The word Alimera derives loosely from the Greek, to mean “day of truth”.c With a commitment to honesty, integrity, responsibility, candor, and trust, Alimera Sciences intend that this promotional information accurately and fairly represents the current state of knowledge of ILUVIEN and DME, and is useful to all healthcare professionals involved in DME and its treatment. a. ILUVIEN SPC. 2013 Available at: www.medicines.org.uk/emc/medicine/27636 (Accessed March 2015) b. Alimera Sciences. Available at www.alimerasciences.com (Accessed March 2015) c. Retina Today. 2011. Available at: retinatoday.com/pdfs/0111RT_Wall.Street.pdf (Accessed March 2015) UK-ILV-MMM-0167 Date of preparation: March 2015 www.alimerasciences.com enquiries@alimerasciences.com

