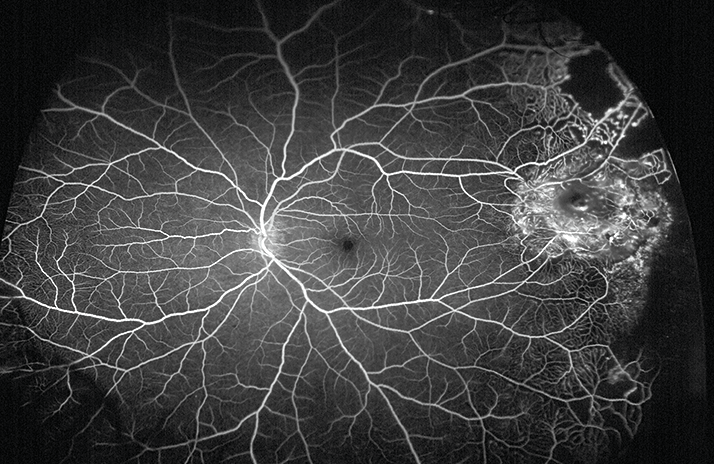
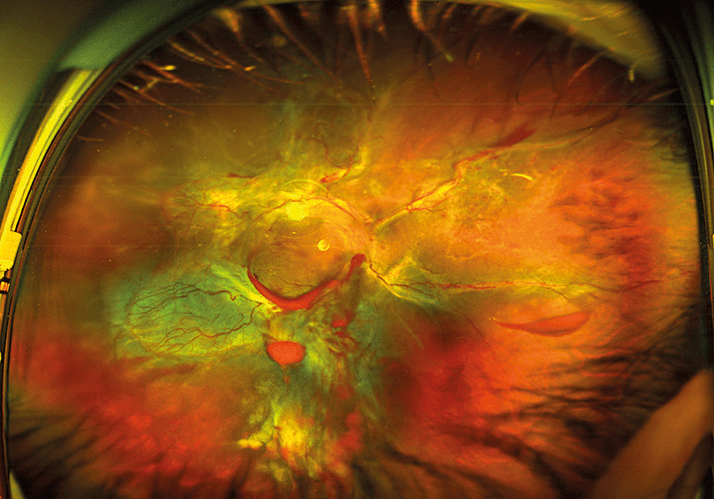
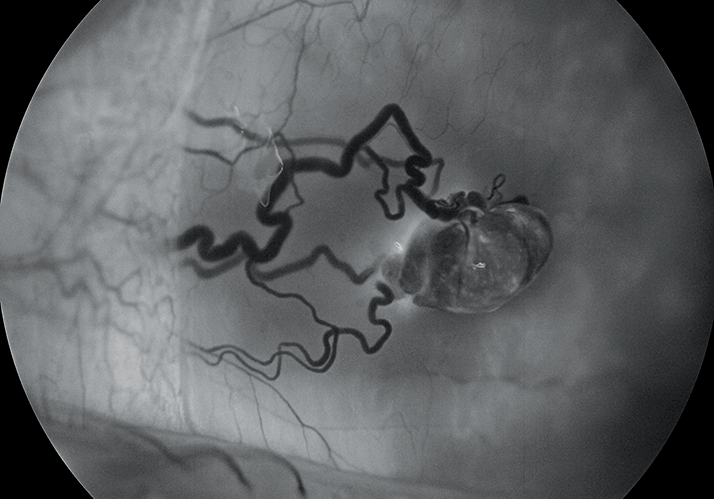
Fluorescein angiography optomap® UWF-image: Coats’ Disease in the far periphery.
Ultra-widefield imaging has become an accepted component in ophthalmic clinical practice demonstrating importance in diagnosis, monitoring and treatment of a wide variety of disease manifestations including diabetic retinopathy, retinal vein occlusion, age-related macular degeneration and inflammatory eye disease.
Carl Glittenberg, CEO of Glittenberg Medical Visualization.
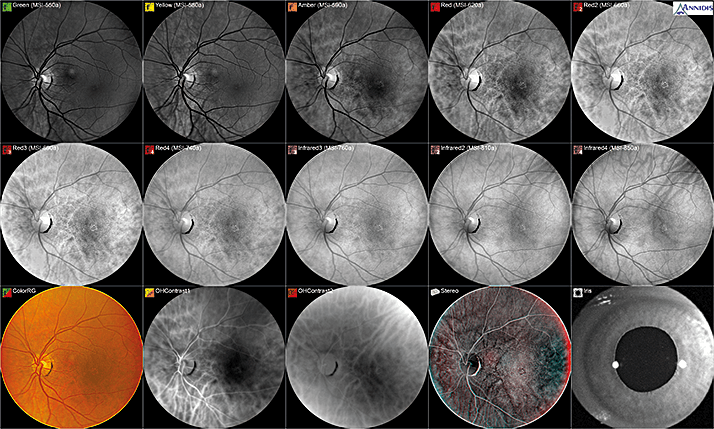
Multispectral imaging (MSI) can reveal more information about a patient’s retina than fundus cameras or scanning laser ophthalmoscopes. This MSI image set (Annidis RHA) shows the changing absorption spectra and spectral sensitivity of retinal structures, as observed in the image sequence of a patient with early RPE disease. The wavelengths beyond 600 nm enhance the visualization of RPE retinal structure and melanin is the dominant retinal pigment. With longer wavelengths, 620 nm to 740 nm (red and deep reds), we can observe window defects within the RPE with a maximum visibility of melanin stacking (dark spots) at 620 nm for this patient without observation of any drusen. The Annidis RHA is a multispectral imaging instrument that utilizes safe light emitting diodes with a wavelength emission range of 500 nm to 850 nm in order to progressively examine the different layers of the retina from the internal limiting membrane to the choroid.
Tim Cole (a); Carl Glittenberg (b–d).
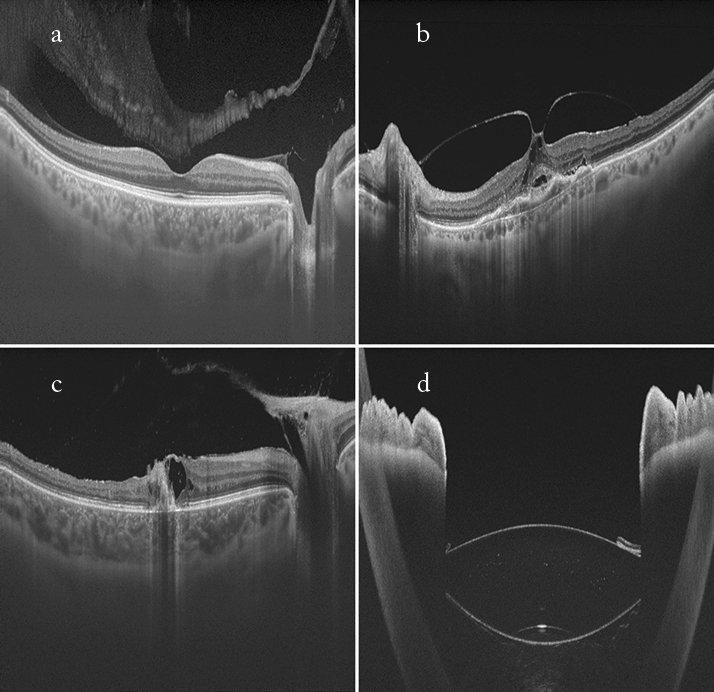
Swept source OCT images (Topcon DRI Atlantis). a. Healthy patient with a visible bursa premacularis in the vitreous above the macula, displaying clear definition of both the choroid and vitreous in one image. b. Vitreomacular traction syndrome (VMTS). Full extent of the VMTS is visible due to large scan width (12 mm). c. Vitreoretinal traction in a patient with posterior uveitis, with clear definition of the choroid and vitreous in one image. Again, the large scan width (12 mm) enabled the full extent of the lesion to be visualized. d. Iris and multifocal IOL: the swept source technology permits the large scan depth visualized.
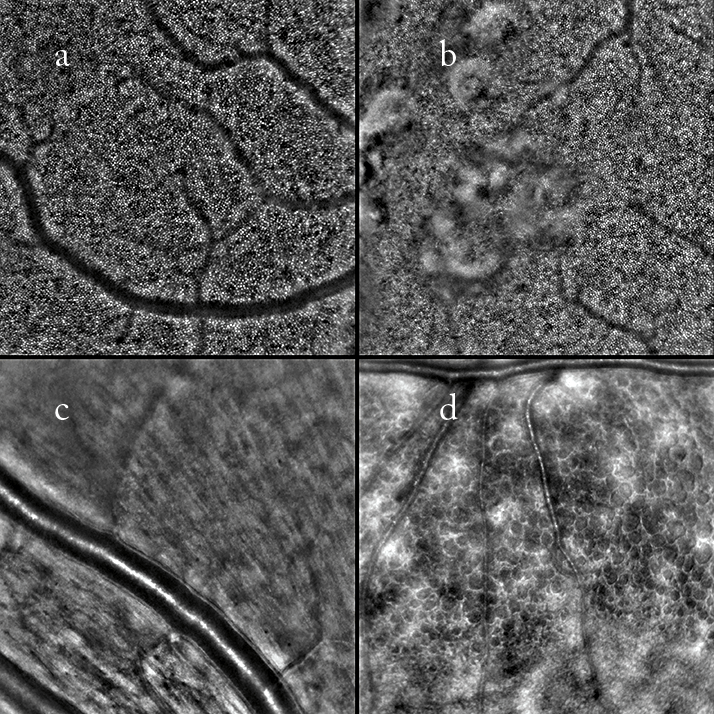
a. Imagine Eyes; b. K . Gocho, V. Sarda, S. Falah, J.A. Sahel, M. Benchaboune, M. Ullern, and M. Paques, Quinze-Vingts National Eye Hospital, Paris, France; c. and d. Dr Kiyoko Gocho, Nippon Medical School Hokusoh Hospital, Chiba, Japan.
Imagine Eyes.
Fundus adaptive optics retinal camera images. a. Cone photoreceptors; b. Reticular drusen; c. Retinal arterioles; d. Microcystic macular edema in a case of autosomal dominant optic atrophy (ADOA).
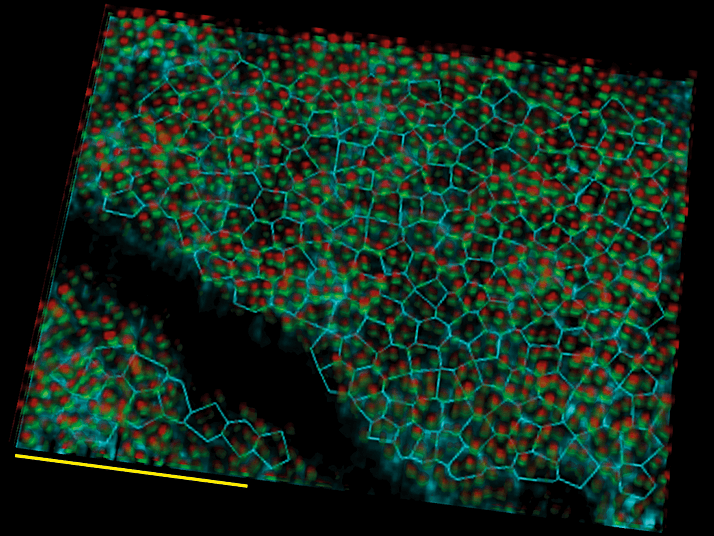
Zhuolin Liu, Omer P. Kocaoglu, Tim L. Turner, and Donald T. Miller, School of Optometry, Indiana University, Bloomington, IN, USA.
3D cellular resolution imaging of the photoreceptor-RPE complex in parafovea of a 48 year old subject, using OCT equipped with adaptive optics. Red and green spots denote reflections from opposing ends of individual cone photoreceptor outer segments. A Voronoi map of the underlying RPE cells (cyan) is superimposed and enables the number of cones per RPE cell to be computed. Scale bar is 100 microns.
Szilard Kiss, Assistant Professor of Ophthalmology, Weill Cornell Medical College, NY, USA.
Ultra-widefield (UWF) imaging is slowly replacing 7-standard field (7-SF) photographs as the standard of care for patients with diabetic retinopathy. Although 7-SF can document the fundus out to approximately 100°, it requires considerable patient cooperation and photographer skill. UWF imaging is able to capture 200° of the retina in a single shot. Here, considerable diabetic pathology (such as areas of neovascularization, hemorrhage and nonperfusion) is noted.
Ultra-widefield (UWF) imaging is slowly replacing 7-standard field (7-SF) photographs as the standard of care for patients with diabetic retinopathy. Although 7-SF is can document the fundus out to approximately 100°, it requires considerable patient cooperation and photographer skill. UWF imaging is able to capture 200° of the retina in a single shot. Here, considerable diabetic pathology (such as areas of neovascularization, hemorrhage and nonperfusion) is noted.
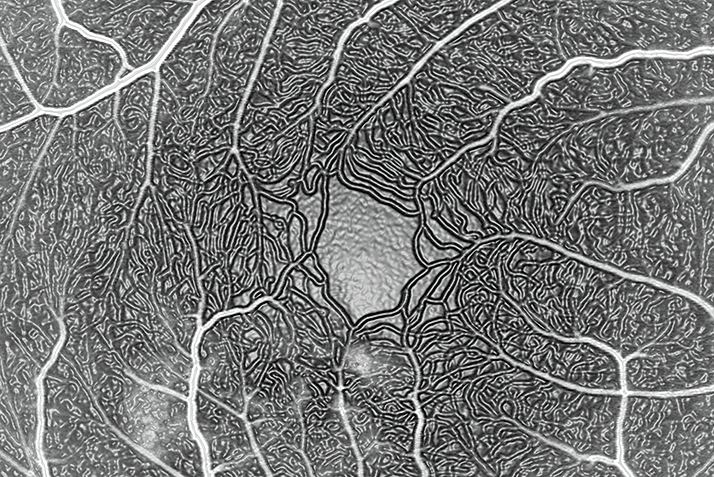
Photographer: Richard Hancock, Assistant Hon.Treasurer of the Ophthalmic Imaging Society, Clinical Sciences, University Hospital Aintree, Liverpool, UK.
A fluorescein angiogram of the right eye in a patient with central serous chorioretinopathy showing leakage. It is very unusual to see the perifoveal network in such fine detail. I have added a Photoshop filter to enhance the retinal vessels (this is not normal practice, but does give a nice effect).
Photographer: Joseph G. Territo, Ophthalmic Imaging Center, Retina Associates of Western New York, Rochester, NY, USA.
Widefield fluorescein angiography is ideal for documenting the complexities of proliferative diabetic retinopathy. It offers the practitioner views into the far peripheral retina where capillary nonperfusion, hemorrhages and neovascularization are often present.

Photographer: Tara Farmer, Imaging Supervisor, Tennessee Retina, Nashville, TN, USA.
Fluorescein angiogram of a pyogenic granuloma at 5 minutes, 40 seconds on a 30-year-old male with a complaint of left eye irritation with no vision loss. The conjunctival hemangioma was 1 mm from plica and 5 mm from the limbus (nasal). The conjunctival lesion was later removed during surgery (excisional biopsy and cryo) and the patient made a full recovery.
Photographer: Michael Singer, Medical Center Ophthalmology Associates, San Antonio, TX, USA.
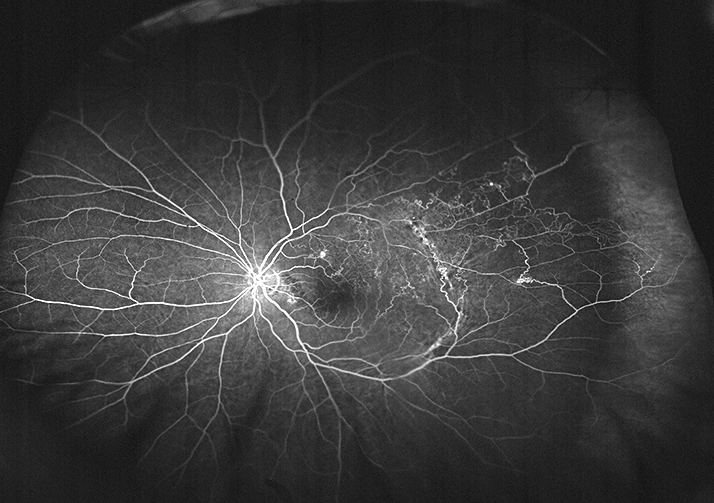
Ultra-widefield fluorescein angiography image demonstrating extensive peripheral changes associated with a retinal vein occlusion including neovascularization, nonperfusion and far peripheral leakage.
Richard Rosen and Toco Chui, Adaptive Optics Imaging Laboratory, New York Eye and Ear Infirmary of Mount Sinai, New York, NY, USA.
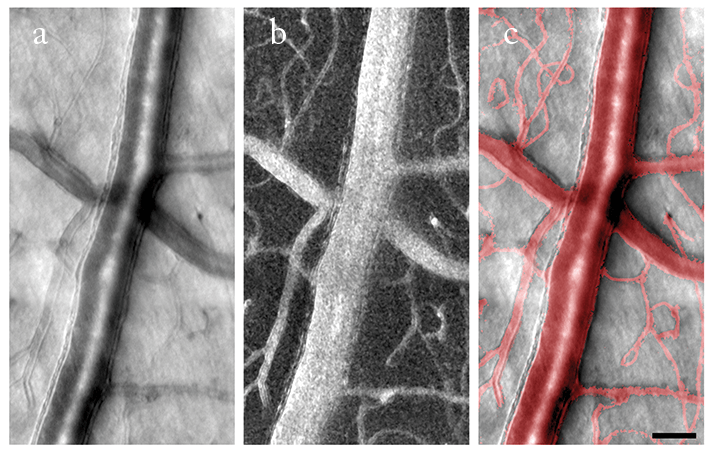
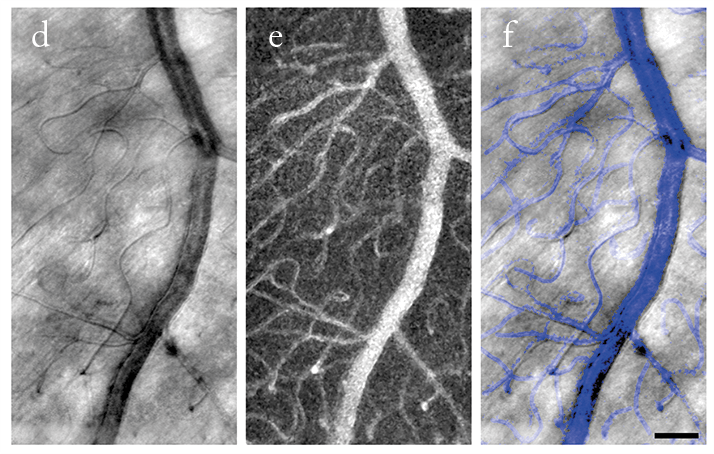
Noninvasive adaptive optics imaging of the retinal arteriole and venule in healthy subjects without the use of an exogenous contrast agent.
a. Adaptive optics image of a 50 μm arteriole located at 5° superior retina. Vascular wall fine structure is readily seen.
b. Corresponding motion contrast image indicates the perfused blood vessels.
c. Superimposition of A and B.
d. Adaptive optics image of a 20 μm venule located at 5° superior retina.
e. Corresponding motion contrast image.
f. Superimposition of D & E.
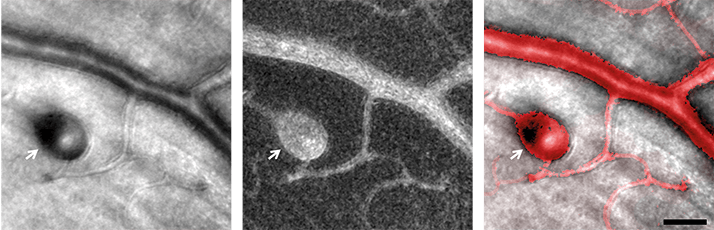
Noninvasive adaptive optics imaging of a retinal microaneurysm (arrows) located at 8° superior retina in a 49-year-old nonproliferative diabetic retinopathy patient. Scale bars, 50 μm.