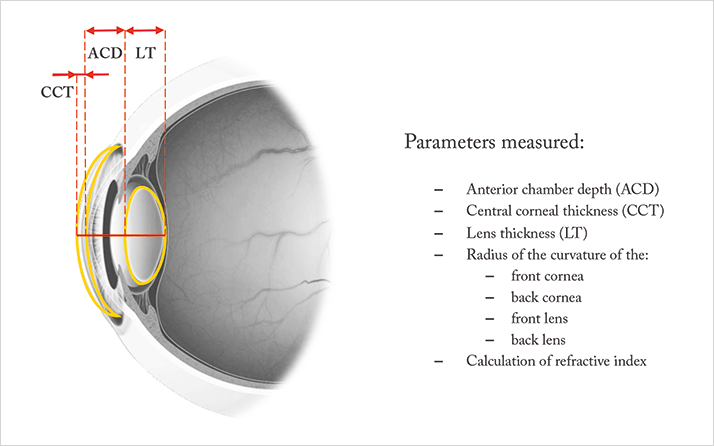
- Surgeons currently have a variety of IOL power formulae at their disposal, but even so, unexpected surgical results are not uncommon
- While working on ray tracing, we were inspired to develop an ocular biometer, Mirricon, that measures every refractive surface in the eye
- Mirricon can calculate lens position and IOL power required without resorting to IOL power formulae
- Our device has just completed an independent and prospective 114-eye trial that has shown it to provide equal or better performance compared with current optical biometers
In the early days of cataract surgery, before the days of A-scan ultrasound axial length biometry, ophthalmologists used a standard 18.0 D prepupillary intraocular lens (IOL) to replace the cloudy crystalline lens they had just extracted – and patients were expected to have the same degree of refractive error after surgery as they had beforehand. But in the 1970s, surgeons began calculating the power of the IOLs they inserted to achieve better vision, based on biometric measurements of the eye – principally, the axial length and keratometry. Many even required “A-constants” – theoretical values specific to the design and placement of individual IOLs. As IOL types diversified and procedures improved, patients began expecting better results from their cataract surgeries. Today many patients demand good vision at both near and far distances, and spectacle independence is the order of the day.
Assumptions and estimations
To accomplish this, surgeons have a variety of different formulae at their disposal to estimate appropriate IOL power. A key part of this estimation is the effective lens position (ELP), which currently relies on the accurate measurement of anterior chamber depth and corneal refractive power, as measured by corneal keratometry or topography. But there are a number of factors and assumptions that can confound this process: each IOL has its own constant that needs to be plugged into the formula, and assumptions are made about the curvature of the posterior corneal surface. Furthermore, if prior refractive surgery has been performed, then IOL power calculators (like the one available online at ASCRS.org) won’t produce a single power recommendation – they will present you with a wide range of options… which is less than ideal. Even without prior refractive surgery, you can make your measurements, follow the rules, use the calculator, and your patient can still experience a “refractive surprise.” Despite the fact that laser refractive surgery results in vision within 0.5 D of the intended target up to 92 percent of the time, fewer than 60 percent of IOL implantations after cataract surgery achieve this goal. Sometimes, there are additional problems like eyes that can’t be measured with the current generation of ocular biometers because of dense cataract, or errors in data entry or transcription.What it comes down to is that, historically, IOL power formulae are good for the “average” refractive outcome, but do a poorer job of predicting individual outcome – especially in eyes with special considerations (such as those that are particularly short or long, astigmatic, or have had previous refractive surgeries, and thus require specific formulae). Understandably, after the effort of determining the best formula to use and then calculating appropriate IOL powers, many surgeons find the relative unpredictability of IOL implantation outcomes frustrating. That’s what drove us to come up with a tool to help eliminate refractive surprises; what we believe to be a better ocular biometer: Mirricon.
A surgeon-led story
The inspiration for Mirricon came from our work on ray tracing for laser refractive surgery. It occurred to us that, if we measured all of the optical surfaces in the eye, we would be able to more accurately estimate the geometrical position of the IOL after implantation. Rather than guessing the lens’ position based on the characteristics of the eye, we thought: why not use data gathered by measuring all of the eye’s optical interfaces to model the ocular tissues? By creating a device capable of such measurements, we wanted to enable ourselves to choose the best IOLs for our patients without having to wrestle with formulae and IOL calculators.To accomplish this, Mirricon uses a new way of combining Purkinje imaging and optical coherence measurements. This combination is not only powerful, but it’s also a completely different technology to anything else currently on the market. Basically, it performs a keratometry-type measurement to assess the topography of each individual surface of the eye (curvatures of the corneal front and back surfaces and lens front and back surfaces, as well as other dimensions of the eye; Figure 1) – something that is pretty unique. When you combine that with an optical coherence measurement system, you gain valuable technical advantages that allow us to minimize error and maximize accuracy.
The story of Mirricon’s creation is unique in that its foundation lies in a surgeon-driven innovation. From the initial inspiration – the confusion of so many different IOL formulae that don’t always provide desirable outcomes – to its ultimate development, the kind of people who are going to use the instrument are the same as the ones developing it: cataract/refractive surgeons.
To the test
After the idea was conceived, Ireland’s National Digital Research Centre in Dublin helped with assembling a team and building the first research system. ClearSight Innovations Ltd was spun out of this collaboration, in order to commercialize the technology and create a prototype that could be brought to clinical trial. It did, and the trial commenced in July of 2014. The trial’s hypothesis was that Mirricon, thanks to its full ray tracing capabilities, is at least equal to or better than current standard-of-care devices. But unlike them, Mirricon doesn’t use formulae or A-constants; instead, it predicts lens position and IOL power from the comprehensive measurements of all of the refractive surfaces of the eye.The trial was a single-site, prospective, observer-masked study, and compared Mirricon with Haag-Streit’s Lenstar optical biometer (Figure 2). From a population of people suitable for monofocal IOL implantation secondary to cataract, 114 eyes in 95 patients were implanted using IOL powers determined by Mirricon. The main objectives of the study were to demonstrate Mirricon’s non-inferiority in predicting postoperative refraction to within 1.0 D of actual three-month outcomes, and in obtaining preoperative measurements in patients’ eyes – both of which were achieved. In fact, the surgeon was actually able to get more eyes to within 1.0 D of target using Mirricon than using the current standard of care – all without using any formulae or IOL power calculators. We were even able to show the superiority of Mirricon to standard-of-care devices in handling patients with astigmatism.
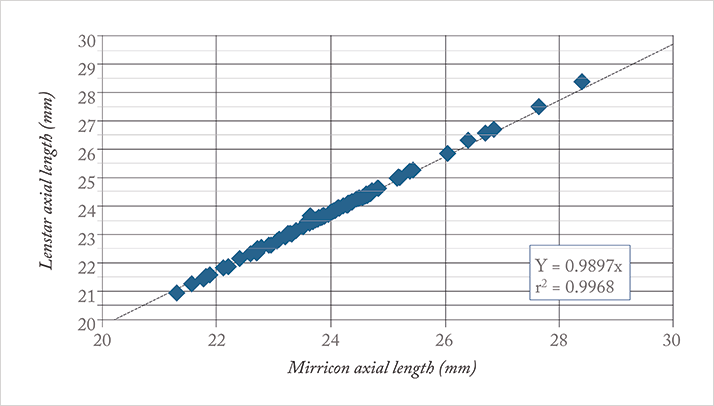
Another important outcome of the study is that, while the Lenstar was only able to measure 95 percent of eyes, Mirricon achieved measurements in all of them – so even in more difficult situations like dense cataracts, where surgeons might normally turn to ultrasound or other options, Mirricon can still measure. Not only did the device provide equal or better performance (while eliminating human transcription errors), it also sped overall workflow in complex patients, as the use of additional devices to perform biometry was not required.
There’s more than one way to skin a cat – and that’s true of ocular biometry, too: Mirricon’s technology is very different from its competitors’. We still have some progress to make; the device is a prototype at the moment, but we’re working to develop a final version. There are more clinical trials in the pipeline, including ones on patients receiving toric IOLs and on post-LASIK patients. Though there’s work to be done, we’re looking forward to providing ophthalmologists with a fast, easy-to-use, surgeon-inspired, ocular biometer and IOL power calculator for use in the clinic.
Michael Mrochen is the founder of IROC Science AG, Zürich, Switzerland, and is co-founder and Chief Technology Officer of ClearSight Innovations (CSI), Dublin, Ireland. Arthur Cummings is a consultant ophthalmologist at the Wellington Eye Clinic and UPMC Beacon Hospital, Dublin, Ireland, and is CSI’s chief clinical advisor. Eugene Ng is a consultant ophthalmologist at the Institute of Eye Surgery, Whitfield Clinic, Waterford, and at the Wellington Eye Clinic in Dublin, Ireland, and holds shares in the NDRC (from which CSI was spun out). Ronan Byrne is co-founder and CEO of CSI.
