
I often hear about companies striving to “meet the unmet need” in ophthalmology. As cliche as this phrase has become, I certainly believe that patients’ needs should be the driving force for innovation in medicine.
Consider the three leading causes of blindness around the world – AMD, cataracts, and glaucoma; despite tremendous progress, we’re still looking for some pieces of each puzzle. There is room for innovation.
The development of intravitreal anti-VEGF injections for wet AMD is one of the best examples of a game-changing technology. Within just a few short years, our ability to preserve driving vision – 20/40 or better – in this patient group has more than quadrupled, and the number of AMD patients designated as legally blind – having vision worse than 20/200 – was halved (1). The challenge we now face is achieving that same level of success in a less burdensome way with extended-release formulations or new drugs with longer duration. At the same time, researchers are rapidly closing in on solutions to the less common, but even more devastating dry form of AMD, with several treatments for geographic atrophy on the horizon.
When it comes to cataracts, we now have a permanent solution that takes less than 10 minutes to perform. However, global healthcare disparities mean that there are still many cases of cataract blindness around the world. In the US, the unmet needs in cataract surgery are now limited to more tightly defined problems, such as consistent management of astigmatism, which may be accomplished through some combination of postoperative lens adjustments, rotationally stable toric IOLs, and better power calculation formulas.
In glaucoma, we have multiple classes of IOP-lowering drugs. The entire MIGS category has also revolutionized our ability to more consistently manage IOP with fewer medications; however, we still lack a fundamental understanding of the underlying mechanisms causing this disease. For patients with glaucoma progression despite IOP lying in the normal range, treatment options are limited.
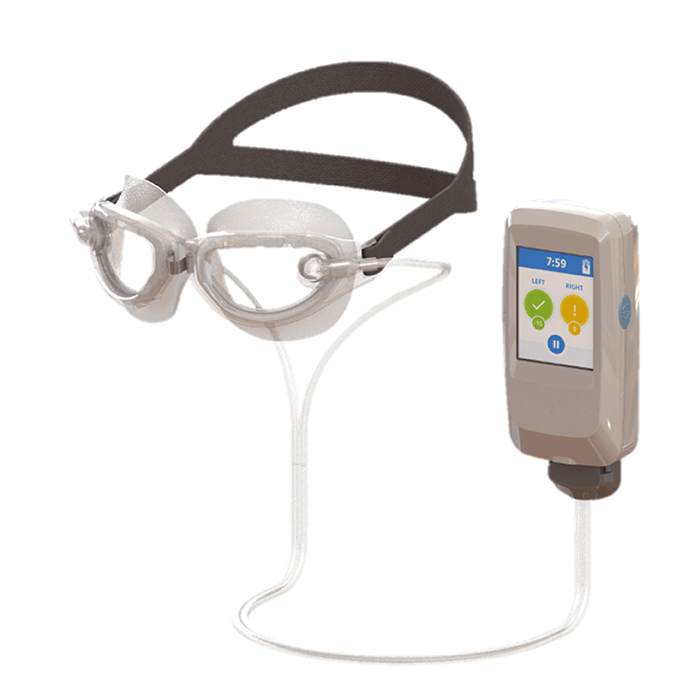
I’m convinced that glaucoma is actually a multi-pressure disease, with IOP being one of the key culprit pressures. Intracranial or cerebrospinal fluid pressure, ocular perfusion pressure, blood pressure, and episcleral venous pressure likely also play a role. Better approaches to lowering IOP and balancing these pressures with a technology like the Multi-Pressure Dial (see Figure 1), may help treat these vulnerable normal-tension glaucoma patients (2). Notably, this concept is not new to medicine; wound vacuums have long been used in the treatment of diabetic foot ulcers and incisional ulcers.
Cell and gene therapies
Ophthalmologists and ophthalmic innovators need to remain aware of – and open to – connections to other specialties that may be the source of the next paradigm shift in innovation. In other cases, innovations in our field can lead to tremendous change across other medical specialties. For example, ophthalmology pioneered the first FDA-approved gene therapy: Luxturna.
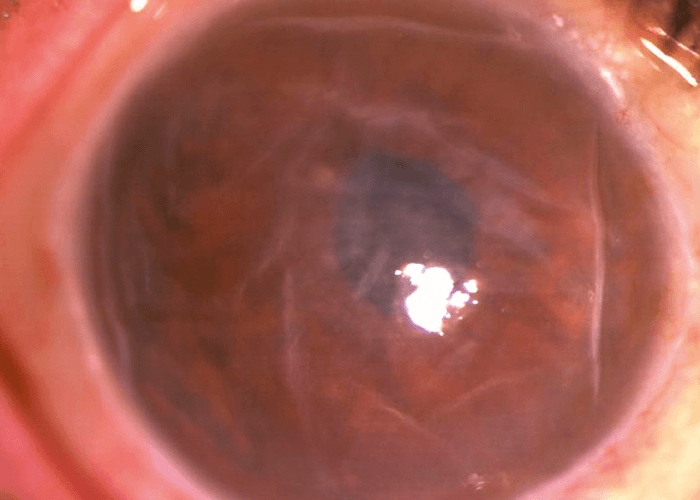
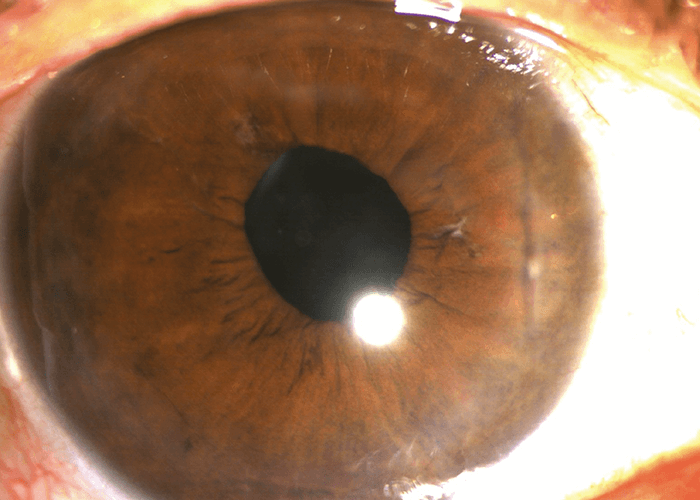
Our specialty is also leading the way in cell therapy thanks to Aurion Biotech and their new approach to addressing corneal blindness from endothelial disease. It is estimated that in the US alone, four percent of the population over the age of 40 suffers from Fuchs endothelial corneal dystrophy, with millions more around the world (3). In addition to Fuchs, pseudophakic corneal edema or bullous keratopathy can lead to decreased vision. Until 10 or 15 years ago, there really was no treatment option until patients’ vision deteriorated enough to require a full-thickness corneal transplant. More recently, advanced endothelial keratoplasties, such as DSEK and DMEK, have improved outcomes, but the reality is that these procedures are technically difficult, limited to subspecialists, and dependent on a limited supply of human donor corneas.
Globally, there is only one cornea available for every 70 eyes with corneal endothelial disease (4).
Aurion’s cultured endothelial cells – from a single donor cornea – can treat hundreds of eyes. First developed by Shigeru Kinoshita at Kyoto Prefecture University School of Medicine in Japan, this technically elegant solution involves the injection of human cultured endothelial cells in a suspension that also includes a rho kinase inhibitor into the anterior chamber of the eye. Results from the company’s recent IOTA trial are consistent with the long-term outcomes reported by Kinoshita for the first 11 eyes he treated (5, 6) where he found the therapy returned patient cortical thickness and visual acuity back to healthy ranges without serious adverse effects. In all, more than 150 procedures have been performed, with truly life-changing results for patients.
Where will the next innovation come from?
In ophthalmology, we are fortunate to have already solved some of the biggest unmet problems patients face, but there are still many “blue sky” opportunities ahead of us. In addition to gene and cell therapies, there are opportunities for innovation in pain control, true accommodating IOLs, artificial intelligence, remote patient monitoring, and the development of treatments for rare conditions, such as retinitis pigmentosa, limbal stem cell deficiency, and acanthamoeba infection.
As ophthalmologists, we have the fascinating advantage of having a direct view into aspects of the human body that aren’t visible in vivo anywhere else – namely, neural tissue in the retina and cellular healing (cell and flare) in the anterior chamber. Using these insights, we have the opportunity to positively impact other areas of medicine.
But a word of warning: As we become more specialized and get better at the task in front of us, we may get worse at finding common ground with other specialties. I encourage my colleagues to be purposeful in learning about innovations in medicine broadly, and to apply those solutions to problems in ophthalmology. Activating those connections enables us to preserve our sense of wonder and curiosity and to be receptive to innovations from any source. The solutions to many of our unmet needs may already be out there somewhere… We just have to connect the dots.
References
- National Eye Institute, “Age-related macular degeneration before and after the era of anti-VEGF drugs” (2016). Available at: https://bit.ly/3zS5sxB.
- TW Samuelson et al., “8 hrs safety evaluation of a multi-pressure dial in eyes with glaucoma: Prospective, open-label, randomized study,” Clin Ophthalmol, 13, 1947 (2019). PMID: 31631962.
- MedlinePlus, “Fuchs endothelial dystrophy” (2018). Available at: https://bit.ly/3biZcoo.
- P Gain et al., “Global survey of corneal transplantation and eye banking,” JAMA Ophthalmol, 134, 167 (2016). PMID: 26633035.
- S Kinoshita et al., “Injection of cultured cells with a ROCK inhibitor for bullous keratopathy,” N Engl J Med, 15, 995 (2018). PMID: 29539291.
- K Numa et al., “Five-year follow-up of first 11 patients undergoing injection of cultured corneal endothelial cells for corneal endothelial failure,” Ophthalmology, 128, 504 (2021). PMID: 32898516.
